A retrospective analysis on the effectiveness of ibuprofen versus conventional dressings in the treatment of diabetic foot ulcers
Received:
Accepted:
Published:
Authors: Afroditi Fotiadou, Kyriacos Evangelou, Georgios Fanariotis, Nikolaos Bekas, Georgios Karaolanis, Konstantinos Vlachos, Dimitrios Chatzis
Conflicts of interest:
The authors declare no conflicts of interest.
Acknowledgements:
Conceptualisation: DC; methodology: DC, GK; software: KE; validation: AF, KE; formal analysis: AF, KE; investigation: GF, NB; resources: GK; data curation: GF, NB; writing — original draft preparation: AF, KE; writing — review & editing: AF, KE, DC; visualisation: AF, KE; supervision: DC, GK; project administration: DC; funding acquisition: DC. All authors have read and agreed to the published version of the manuscript.
Funding:
This study did not receive any funding.
Statements of consent and ethical approval:
All authors adhered to the principles of the Declaration of Helsinki as far as the ethical principles for medical research involving human subjects are concerned. Informed consent was obtained from all patients who participated in the study.
Corresponding author:
Afroditi Fotiadou, Resident Surgeon, Doktorantin, Ruprecht Karls University Heidelberg, Im Neunheimer Feld 420, 69120, Heidelberg, Germany. Email: aphroditefotiadou@gmail.com, Afroditi.Fotiadou@med.uni-heidelberg.de
Surgical debridements and limb amputations are routinely performed for diabetic foot ulcers but predispose to pain and infection. This study aimed to evaluate the impact of ibuprofen dressings on healing and quality of life in diabetic foot ulcers postoperatively. A four-year retrospective analysis was conducted at a university hospital. Patients received Ibuprofen, vacuum-assisted closure devices, foam, foaming silicone, conventional, collagen, or hydrofiber dressings. Bed rest duration, length of stay, healing time, pain and distress scores were recorded. Statistical analysis included Mann-Whitney U, Pearson chi-squared, Kruskal-Wallis H, Fisher-Freeman-Halton exact tests, and ANCOVA with Holm-Bonferroni-corrected post hoc testing were performed. All p-values are two-tailed and statistical significance set at p<0.05. The cohort (217 patients) had a mean age of 72.8 ± 11.3 years, with 127 limb amputations and 90 surgical debridements. Foaming silicone dressings prevailed (30.0%) and local anaesthesia was preferred for limb amputations (88.2%) over surgical debridements (1.1%; p<0.001). Single-toe amputations (49.6%) and abscess drainage with debridement (56.7%) predominated. Mean pain score and distress score were low (3.8 ± 1.7) and moderate (11.0 ± 3.0) in limb amputations but both moderate in surgical debridements (10.3 ± 2.9 and 13.9 ± 3.0, respectively). Bed rest duration, length of stay, healing time, and pain and distress scores were improved in limb amputations (p<0.001), while Ibuprofen also demonstrated shorter bed rest duration and length of stay than conventional dressings closure devices (p<0.001). Vacuum-assisted devices had the lowest pain score among all dressings (p≤0.010). For surgical debridements, Ibuprofen was observed to promote shorter bed rest duration than foaming silicone dressings (p=0.026) and lower distress score than vacuum-assisted devices (p=0.028). To conclude, Ibuprofen dressings were associated with shorter bed rest duration and hospital stay compared to conventional dressings, though results versus advanced dressings such as VAC or foam silicone were less consistent. Differences in healing time did not consistently reach statistical significance across all comparisons. Further studies are necessary to validate these findings and evaluate long-term outcomes.
Diabetic foot ulcers (DFUs) are a major public health issue that affects 25–50% of patients with diabetes worldwide. DFUs are the reason for 50–70% of lower extremity amputations and the primary indication for non-traumatic amputation, increasing hospital care costs up to 10-fold (McDermott et al, 2023). Severe infection and peripheral vein necrosis are the most common complications, with multiple psychological, social and cultural repercussions (Niță et al, 2023). The Wagner-Meggitt classification is the most widely used classification for DFUs, categorising them into six grades (0-5) based on their location, size, depth, extent, and the presence of infection and/or gangrene (Wang et al, 2022). Several other frameworks are used. These include the University of Texas (UT) staging system, the Perfusion, extent, depth, infection, and sensation (PEDIS) score, and the Site, ischaemia, bacterial infection, area, and depth (SINBAD) classification. Furthermore, the International Working Group on the Diabetic Foot (IWGDF) provides evidence-based guidelines on the prevention and management of diabetes-related foot disease (Santema et al, 2016).
The treatment of patients with diabetic foot ulcers is challenging due to the pathophysiological complexity of diabetes and individual patient characteristics, needs, and comorbidities (Doğruel et al, 2022). Surgical debridements and extremity amputations are the main surgical treatment and nowadays are regarded as routine procedures in vascular surgery departments; however, they often increase the risk of postoperative infection and morbidity (Norman et al, 2022). The following four types of surgical debridements are mostly used:
- Surgical debridement of the calf.
- Foot abscess drainage.
- Foot abscess drainage with toe amputation.
- Foot abscess drainage with surgical debridement.
Forefoot amputations are traditionally classified into seven levels (Dayya et al, 2022):
- Amputation of a single toe with or without the corresponding metatarsal head.
- Amputation of two toes with or without the corresponding metatarsal heads.
- Amputation of three toes with or without the corresponding metatarsal heads.
- Amputation of more than three toes with or without the corresponding metatarsal heads.
- Transmetatarsal amputation.;
- Tarsometatarsal disarticulation (Lisfranc amputation).
- Transtarsal (Chopart) amputation.
Wound dressings have emerged as a valuable means of postoperative targeted drug delivery and localised pain relief over the last vicennium, as most gold standard surgical treatments mandate subsequent revascularisation. The various dressings used in everyday clinical practice include activated carbon (charcoal), with silver salts for lesions with a large amount of exudate; hydrocolloids in small and medium lesions; antibiotics and collagenase. Additionally, topical drugs are often recommended for their localised impact on nearby tissues, as they effectively target areas surrounding the application site (Nguyen et al, 2023; Oliveira et al, 2023). Regarding non-steroidal anti-inflammatory drugs (NSAIDs), ibuprofen is progressively gaining ground thanks to its analgesic and anti-inflammatory properties (Palao i Domenech et al, 2008; Saco et al, 2016; Cavalcanti et al, 2017; Lee et al, 2017; Güiza-Argüello, 2022; Ffrench et al, 2023; Nguyen et al, 2023; Oliveira et al, 2023; Drosopoulou et al, 2025).
While there are promising results on the benefits of ibuprofen dressings in patients with DFUs, the literature contains limited evidence from large patient cohorts. On this basis, the authors present their experience in treating patients with DFUs using various types of dressings, aiming to:
- Evaluate the effect of ibuprofen dressings on quality of life improvement.
- Compare ibuprofen dressings with other types of commonly used dressings, including plain dressings, gauzes and antiseptic ointments.
Materials and methods
Design
This single-centre retrospective study was carried out between July 2016 and August 2020 at the vascular surgery department of Ioannina University Hospital, Ioannina Greece. The study population comprised inpatients with painful, gangrenous DFUs who underwent either amputation in cases of an open non-exudative wound (dry gangrene), or extensive necrotic tissue debridement (often with concurrent underlying bone removal) in cases of septic wounds (wet gangrene). The allocation of patients to dressing type was not randomised, but rather based on clinician preference, wound characteristics (exudate amount, presence of infection, location and size) and dressing availability during the study period. Patients with moderate to severe exudate who were capable of self-evaluating pain intensity and relief levels were included. The study was conducted in accordance with the Declaration of Helsinki II, the EC medical device directive (EC, 1993) and the international standard on clinical investigation of medical devices for human subjects (ISO, 2003). Ethics committee approval was granted, and written informed patient consent obtained.
Operations were performed by vascular, orthopaedic, plastic-reconstructive and general surgeons. The study employed the following seven dressing types, based on routine use and availability in the vascular surgical department:
- Vacuum-assisted closure (VAC) devices.
- Foam dressings (FOAM).
- Foaming silicone dressings (FOAS).
- Ibuprofen dressings (IBU).
- Collagen dressings (COL).
- Hydrofiber dressings (HFIB).
- Conventional dressings (CONV), including gauzes, betadine and hydrogen peroxide.
Data collection and preparation
All patients who underwent amputation for a persisting open wound and/or extensive surgical debridement for diabetic or septic foot between July 2016 and August 2020 were included in a cohort. The vast majority of patients underwent a reperfusion intervention (either open, endovascular or hybrid). Conservative or surgical treatment followed surgeon experience and preference. The seven types of amputation and four types of surgical debridement performed were as previously described. Anaesthesia was administered according to an agreed plan and included local, regional or general anaesthetics, depending on the complexity and duration of the procedure, as well as individual patient needs.
Healing time (HT) was defined as the interval between the day the amputation or surgical debridement was performed and the first documented date of complete healing, as determined by the attending specialist surgeon. HT, bed rest duration (BRD) and length of stay (LOS) were recorded in days. A pain score (PS) leaflet completed by the treating physician was designed to quantify patient pain levels and determine analgesia needs (minimum score: 0; maximum score: 18). A self-completion distress score (DS) leaflet [Figure 1] was also distributed to evaluate patient discomfort throughout the hospital stay (minimum score: 4; maximum score: 20).


Figure 1. Pain score leaflet. Figure 2. Distress score leaflet.
Patients were followed up for a minimum of 48 months after treatment ended, and complications were defined as minor or major according to the Clavien-Dindo classification (Frykberg and Banks, 2015). Demographic and clinical information and data were archived after every discharge and stored in institutional electronic files, which were re-opened for the purpose of the study.
Statistical analysis
Quantitative variables were expressed as means with standard deviations, and medians with ranges; qualitative variables were expressed as absolute and relative frequencies.
For the comparison of quantitative variables between two groups (type of intervention), the Kolmogorov-Smirnov and Shapiro-Wilk goodness-of-fit tests were used to ascertain whether the data were normally distributed among both groups, and the Mann-Whitney U test to compare the means between patients who underwent amputation or surgical debridement.
For the comparison of qualitative variables between two groups (type of intervention), Pearson’s chi-squared test of independence was used to evaluate the existence of dependence between subgroups.
For the comparison of quantitative demographic variables between more than two groups (type of dressing), the Shapiro-Wilk goodness-of-fit test was used to ascertain whether the data was normally distributed among all seven groups, and the Kruskal-Wallis H (one-way ANOVA on ranks) test to compare the mean age between patients who were treated with different types of dressings.
For the comparison of qualitative demographic variables between more than two groups (type of dressing), the Fisher-Freeman-Halton exact test was used to evaluate the existence of dependence between subgroups.
For the comparison of quantitative outcome variables (BRD, HT, LOS, PS, and DS) between more than two groups (type of dressing), Levene’s test for equality of variances, and one-way analysis of covariance (ANCOVA) to compare the means between patients who received treatment with different types of dressings, with all available demographic variables (age, type of operation, and type of anaesthesia) added as covariates to a fixed-effects model.
For the comparison of qualitative variables (death during treatment) between more than two groups (type of dressing), a binary logistic regression model was developed to investigate the association between the type of dressing and the risk of death during treatment, while controlling for age, type of operation, and type of anaesthesia.
All reported p-values are two tailed. Statistical significance was set at p<.05, and analysis was conducted using IBM SPSS Statistics for Windows (version 29.0.2.0; Boulton et al, 2018).
Results
Overall patient characteristics
The study included 217 patients, with 127 undergoing amputation and 90 subjected to extensive surgical debridement. Seven transtibial amputations and four transfemoral amputations with residuum infection were also treated.
The demographic and clinical characteristics, both overall and categorised intervention type, for the 127 patients in the cohort are summarised in Table 1. Mean patient age was 72.8 ± 11.3 years and FOAM dressings were the most prevalent (30.0%). Interestingly, local anaesthetics predominated in amputations (88.2%) while surgical debridements were mostly performed under regional (76.7%) or general (22.2%) anaesthesia.
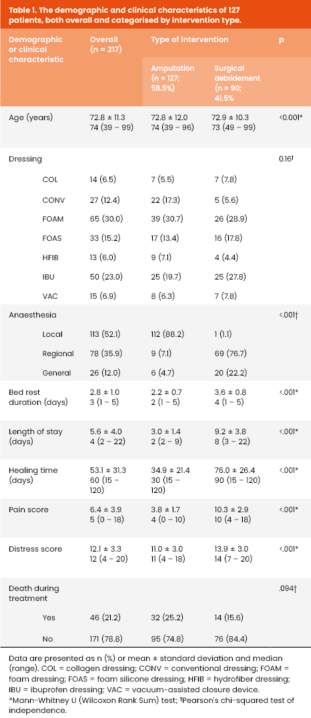
The type of dressing used did not differ significantly between the two surgical procedure groups. Almost all patients in the amputation group received local anaesthesia, while almost 99% of patients who underwent debridement received either regional or general anaesthesia (p<0.001).
Mean BRD, LOS, and HT were significantly longer in the surgical debridement group by 1.4 days, 6.2 days, and 41.1 days, respectively (p<0.001). Regardless of the type of dressing used, healing lasted less than 2 months in 37.8% of patients. The mean PS and DS were also higher in the debridement group by 6.5 and 2.9, respectively (p<0.001). However, the overall mortality rate was higher among patients undergoing amputation (25.2% versus 15.6%).
Amputations
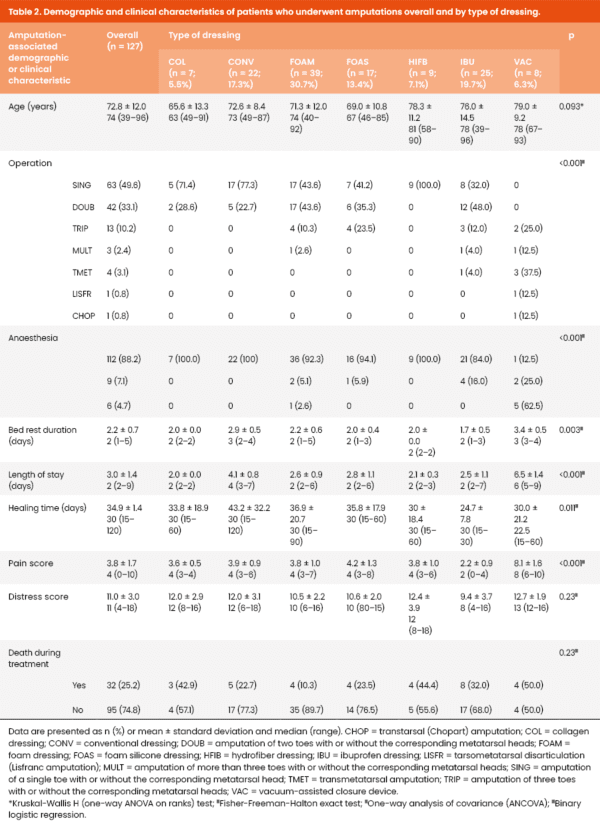
The demographic and clinical characteristics of patients who underwent amputations, overall and by type of dressing are summarised in Table 2. The mean patient age was 72.8 ± 12.0 years and amputations of a single toe with or without the corresponding metatarsal head were the most common (49.6%). Hydrofiber dressings were only used for the above amputation type, while tarsometatarsal (Lisfranc) disarticulations and transtarsal amputations were always dressed using VAC devices. Collagen and conventional dressings were also used for amputations of a single toe or two toes with or without the corresponding metatarsal heads. Mean PS was low (3.8 ± 1.7), although the mean DS was higher and relatively moderate (11.0 ± 3.0).
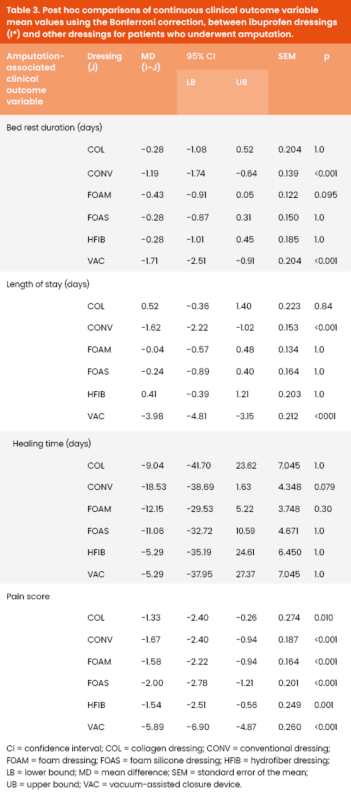
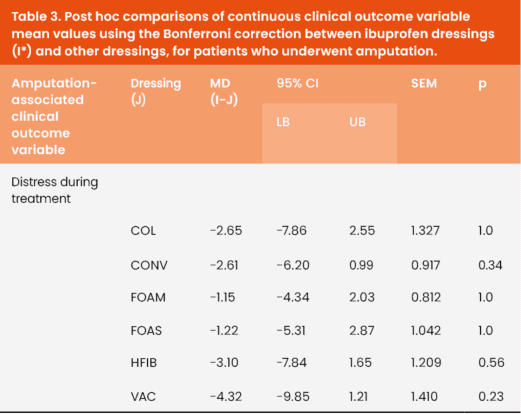
Clinical outcome variable mean values between ibuprofen dressings and other dressings, for patients who underwent amputation, are presented in Table 3, alongside the results of the post hoc testing.
Bed rest duration
A one-way ANCOVA revealed a statistically significant difference in mean BRD between the different dressing types [F(6,11) = 6.878, p=0.003], while adjusting for age, type of operation and type of anaesthesia. The partial eta-squared value (η2 =0.790) indicates that the effect size was large.
Post hoc comparisons with Bonferroni correction indicated that IBU dressings were associated with a shorter BRD compared to CONV (MD=−1.19 days, SEM=.139, p<0.001) and VAC (MD=−1.71, SEM=0.204, p<0.001) dressings. The mean difference in BRD between IBU and the rest of dressings was not statistically significant.
Length of stay
A one-way ANCOVA revealed a statistically significant difference in the mean LOS between the different types of dressings [F(6,11) = 11.402, p<.001] while adjusting for age, type of operation, and type of anaesthesia. The partial eta-squared value (η2 = .861) indicates that the effect size was large.
Post-hoc comparisons with Bonferroni correction indicated that IBU dressings were associated with a shorter LOS compared to CONV (MD=-1.62 days, SEM=.153, p<.001) and VAC (MD=-3.98, SEM=.212, p<.001) dressings. The mean LOS difference between IBU and the rest of dressings was not statistically significant.
Healing time
A one-way ANCOVA revealed a statistically significant difference in mean HT between the different types of dressings [F(6,7) = 6.976, p=.011] while adjusting for age, type of operation, and type of anaesthesia. The partial eta-squared value (η2 = .857) indicates that the effect size was large.
However, post hoc comparisons with Bonferroni correction indicated that the mean difference in HT between IBU and the rest of dressings was not statistically significant.
Pain score
A one-way ANCOVA revealed a statistically significant difference in mean PS between the different types of dressings [F(6,11) = 11.715, p<.001] while adjusting for age, type of operation, and type of anaesthesia. The partial eta-squared value (η2 = .865) indicates that the effect size was large.
Post-hoc comparisons with Bonferroni correction indicated that IBU dressings were associated with better mean PS compared to all other dressings; COL (MD=-1.33, SEM=.274, p=.010); CONV (MD=-1.67, SEM=.187, p<.001); FOAM (MD=-1.58, SEM=.164, p<.001); FOAS (MD=-2.00, SEM=.187, p<.001); HFIB (MD=-1.54, SEM=.249, p=.001); VAC (MD=-5.98, SEM=.260, p<.001).
Distress score
A one-way ANCOVA revealed no statistically significant difference in mean DS between the different types of dressings [F(6,11) = 1.612, p=.233] while adjusting for age, type of operation, and type of anaesthesia.
Post-hoc comparisons with Bonferroni correction confirmed that the mean difference in DS between IBU and the rest of the dressings was not statistically significant.
Death during treatment
The overall binary logistic regression model explained 23.4% of the variation of death during treatment (Nagelkerke R2), and correctly predicted 77.2% of cases. The association between the type of dressing used and the risk of death during treatment was not statistically significant (p=.23).
Surgical debridement
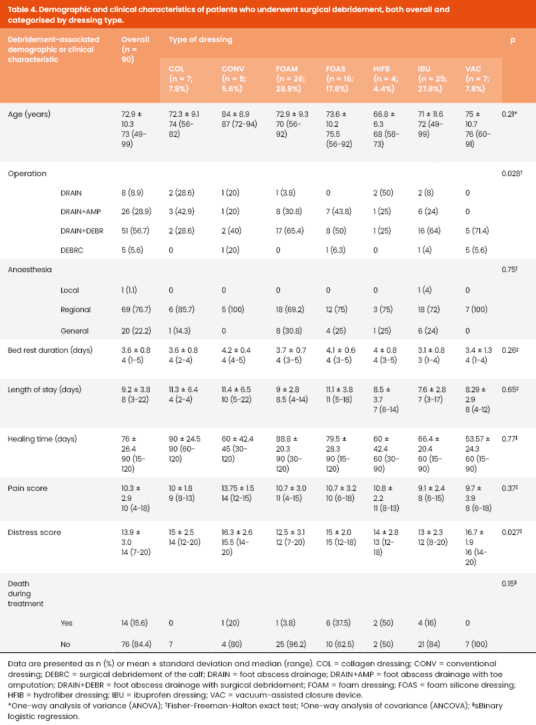
The demographic and clinical characteristics of patients who underwent surgical debridement, overall and by type of dressing, are summarised in Table 4. The mean age was 72.9 ± 10.3 years. Ibuprofen and foam dressings were the most frequently used (27.8% and 28.9%, respectively). Most patients underwent foot abscess drainage with surgical debridement (56.7%); regional anaesthesia was the most common choice (76.7%) and was used in all cases subsequently treated with VAC devices. Although the overall DS were again moderate (mean: 13.9 ± 3.0), the PS was also moderate (10.3 ± 2.9), contrary to the low PS in amputations.
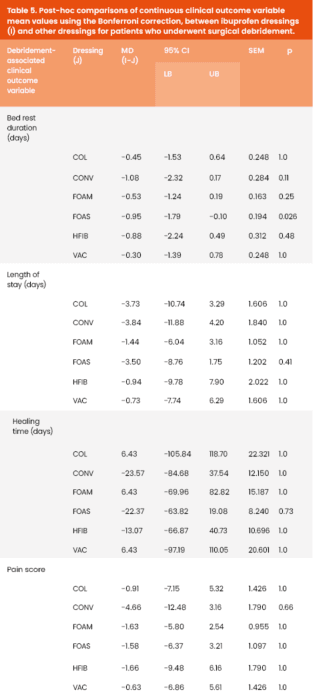
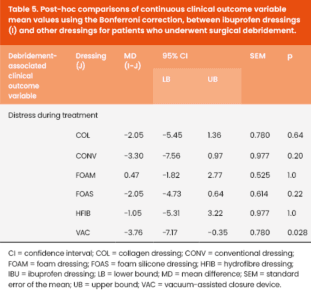
Mean values for clinical outcome variables, comparing ibuprofen dressings with all other dressing types for patients who underwent surgical debridement, are presented in Table 5, alongside the results of the post-hoc testing.
Bed rest duration
A one-way ANCOVA revealed no statistically significant difference in the mean BRD between the different types of dressings [F(5;8) = 1.632, p = .26] while adjusting for age, type of operation, and type of anaesthesia.
However, post-hoc comparisons with the Bonferroni correction indicated that IBU dressings were associated with a shorter BRD compared to FOAS dressings (MD = -.95 days, SEM = .194, p=.026). The mean difference in BRD between IBU and the rest of dressings was not statistically significant.
Length of stay
A one-way ANCOVA revealed no statistically significant difference in the mean LOS between the different types of dressings [F(5,8) = .675, p = .66] while adjusting for age, type of operation, and type of anaesthesia.
Post-hoc comparisons with Bonferroni correction confirmed that the mean difference in the LOS between IBU and the rest of dressings was not statistically significant.
Healing time
A one-way ANCOVA revealed no statistically significant difference in mean HT between the different types of dressings [F(5,6) = 0.772, p=.60] while adjusting for age, type of operation, and type of anaesthesia.
Post-hoc comparisons with Bonferroni correction confirmed that the mean difference in HT between IBU and the rest of dressings was not statistically significant.
Pain score
A one-way ANCOVA revealed no statistically significant difference in mean PS between the different types of dressings [F(5,8) = 1.248, p=0.37] while adjusting for age, type of operation, and type of anaesthesia.
Post hoc comparisons with Bonferroni correction confirmed that the mean difference in PS between IBU and the rest of dressings was not statistically significant.
Distress score
A one-way ANCOVA revealed no statistically significant difference in mean DS between the different types of dressings [F(5,8) = 4.684, p=0.027] while adjusting for age, type of operation, and type of anaesthesia.
Post hoc comparisons with Bonferroni correction confirmed that IBU dressings were associated with better mean DS compared to VAC devices (MD = -3.76, SEM =0.78, p=0.028). The mean difference in DS between IBU and the rest of dressings was not statistically significant.
Death during treatment
The overall binary logistic regression model explained 42.6% of the variation of death during treatment (Nagelkerke R2), and correctly predicted 83.3% of cases. The association between the type of dressing used and the risk of death during treatment was not statistically significant (p=0.153). Detailed death during treatment rates by dressing type are presented in Tables 2 and 4.
Discussion
More than 60% of patients with DFUs report severe wound pain (Boulton et al, 2018). Chronic and painful ulcers frequently stall in an inflammatory phase where excessive exudate damages healthy periwound skin, impedes healing and prolongs overall treatment times (Frykberg and Banks, 2015). Sleep disturbances, mobility restriction, daily activity limitation and quality of life deterioration are among the most common side effects (Cavalcanti et al, 2017). Two key types of wound-associated pain are broadly recognised, with neuropathic pain arising from nerve tissue damage and nociceptive pain being the result of actual tissue damage (Lindholm et al, 2019; Finnerup et al, 2021). Although oral non-steroidal anti-inflammatory drugs (NSAIDs) are known to reduce nociceptive pain, they are associated with gastrointestinal bleeding and renal function impairment. Systemic NSAIDs may be less effective in patients with circulatory compromise. In contrast, topical ibuprofen achieves therapeutic levels at the wound site with lower overall doses, fewer systemic side effects, and reduced dependency on local circulation (Tai and McAlindon, 2021).
Although spontaneous open wound improvement and medically-induced healing are possible, surgical or intravascular intervention remain the gold standard of treatment. As the two most popular surgical treatment techniques (amputation and debridement) often exacerbate postoperative pain and predispose to infections, wound dressings are progressively adopted more widely by the global vascular surgery community for inflamed and infected open wounds (Frykberg and Banks, 2015). There are emerging indications that ibuprofen dressings, alone or in combination with regular monitoring and thorough wound care, can minimise inflammation and alleviate pain, allowing for faster healing and mobilisation and ultimately improving mobility, sleep and mood (Price et al, 2007; Palao et al, 2008; Romanelli et al, 2009; Salomé and Ferreira, 2017).
Ibuprofen is a NSAID that inhibits cyclooxygenase (COX1 and COX2) enzymes; these enzymes convert arachidonic acid to prostaglandin H2 (PGH2), thereby desensitising peripheral nerve endings that transmit pain signals from inflamed areas (Price et al, 2007). Topical ibuprofen administration with direct drug delivery achieves therapeutic levels within target-tissues while sparing plasma and obviating systemic side effects. Direct on-site application provides adequate tissue penetration and achieves straightforward analgesia in the affected areas. In addition, Ibuprofen dressings are convenient and easy-to-use, improving patient compliance and satisfaction and minimising the risk of iatrogenic errors. Several types of ibuprofen dressings are available, including gels that are easy to apply and appropriate for a variety of wounds; patches that provide long-lasting pain relief; foam dressings for moist wound healing; and impregnated dressings (Price et al, 2007; Romanelli et al, 2009; Salomé and Ferreira, 2017).
Two trials by Arapoglou et al (2011) and Gottrup et al (2007) evaluated the effects of slow-release ibuprofen foam dressings on persistent venous leg ulcer pain. The studies included 470 patients who were compared to individuals managed with standard treatment. Significantly more participants in the ibuprofen dressing group achieved the desired outcome of >50% improvement in the total maximum pain relief score between day 1 and day 5 than those in the standard treatment group (RR 1.63; 95% CI = 1.24 - 2.15; p=.0006). The researchers concluded that ibuprofen-containing dressings can be a safe and effective pain relief option (Gottrup et al, 2007; Arapoglou et al, 2011).
What distinguishes our analysis from previous studies is that we present the largest to date patient cohort comparing IBU dressings with all other dressing types available in everyday clinical practice, and simultaneously evaluate PS and DS. We observed optimal results in terms of pain and discomfort following lower extremity amputation with IBU dressings, as well as superior similar outcomes compared to most dressings. This study though, is limited by its retrospective design, which precludes randomisation and introduces selection bias, as dressing allocation depended on clinician judgment, wound characteristics and product availability. Furthermore, potential confounders such as infection severity, vascular status, comorbidities, use of antibiotics, and previous revascularisation procedures were not systematically recorded or controlled for, which may have influenced outcomes.
Furthermore, our analysis suggests that ibuprofen dressings may offer advantages compared to conventional dressings in terms of length of stay and pain management. However, these differences were not always statistically significant, and benefits were less pronounced when compared with advanced dressings such as VAC or foam silicone. This could be attributed to the relatively limited sample size of our cohort, which may have reduced statistical power, and the potential interference of other factors such as unmeasured comorbidities. Another important limitation is that the release of ibuprofen from the dressings depends on the presence of sufficient wound exudate. In this retrospective analysis, the level of wound exudate was not systematically documented; therefore, its potential effect on ibuprofen bioavailability and clinical outcomes could not be assessed. This factor may partly explain the variability in our findings. Therefore, our results should be interpreted with caution, and no causal relationship can be established. The apparent benefit observed in certain subgroups may also reflect confounding factors such as wound characteristics or comorbidities (e.g. concurrent medications and infection severity). Considering that the various dressings have comparable costs, IBU dressings can be considered a comprehensive approach for open DFU management (Oliveira et al, 2023).
Although beneficial, IBU dressings are not suitable for all patients; those allergic to ibuprofen or other NSAIDs, and/or with pre-existing conditions such as renal or liver failure, are at an increased risk of NSAID-induced side effects and should be managed with great caution under clinical physician supervision. Healthcare providers should evaluate the risks and benefits of ibuprofen dressings in each individual case, carefully monitor patients and consider alternative treatment options for those with cardiovascular and/or renal disease.
Conclusion
Ibuprofen dressings can be a valuable treatment option for diabetic foot ulcers, offering targeted drug delivery and localised pain relief. This study demonstrated that IBU dressings may provide benefits in terms of pain and discomfort in patients with DFUs, with healing time and length of stay comparable to conventional dressings. Nevertheless, further and larger studies are necessary to confirm these findings and evaluate long-term outcomes. Ultimately, selecting the appropriate wound dressing can expedite healing and significantly reduce both patient morbidity and the financial burden on healthcare services for those with DFUs.
References
Arapoglou V, Katsenis K, Syrigos KN, et al (2011) Analgesic efficacy of an ibuprofen-releasing foam dressing compared with local best practice for painful exuding wounds. J Wound Care 20(7): 319–325. doi: 10.12968/jowc.2011.20.7.319
Boulton AJM, Armstrong DG, Kirsner RS, et al. Diagnosis and management of diabetic foot complications. American Diabetes Association, Arlington, VA
Cavalcanti LM, Pinto FCM, Oliveira GM, et al (2017) Efficacy of bacterial cellulose membrane for the treatment of lower limbs chronic varicose ulcers: a randomized and controlled trial. Rev Col Bras Cir 44(1): 72–80. doi: 10.1590/0100-69912017001011
Clavien PA, Barkun J, de Oliveira ML, et al (2009) The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg 250(2): 187–96. doi: 10.1097/SLA.0b013e3181b13ca2
Dayya D, O'Neill OJ, Huedo-Medina TB, et al (2022) Debridement of diabetic foot ulcers. Adv Wound Care (New Rochelle) 11(12): 666–86. doi: 10.1089/wound.2021.0016
Doğruel H, Aydemir M, Balci MK (2022) Management of diabetic foot ulcers and the challenging points: an endocrine view. World J Diabetes 13(1): 27–36. doi: 10.4239/wjd.v13.i1.27
Drosopoulou K, Kosheleva RI, Ofrydopoulou A, et al (2025) Topical and transdermal delivery of nonsteroidal anti-inflammatory drugs (NSAIDs) for inflammation and pain: current trends and future directions in delivery systems. Processes 13(3): 907. doi: 10.3390/pr13030907
EC (2003) Council Directive 93/42/EEC of 14 June 1993 concerning medical devices. Version 2003-03-27.
Ffrench C, Finn D, Velligna A, et al (2023) Systematic review of topical interventions for the management of pain in chronic wounds. Pain Rep 8(5): e1073. doi: 10.1097/PR9.0000000000001073
Finnerup NB, Kuner R, Jensen TS (2021) Neuropathic pain: from mechanisms to treatment. Physiol Rev 101(1): 259–301. doi: 10.1152/physrev.00045.2019
Frykberg RG, Banks J (2015) Challenges in the treatment of chronic wounds. Adv Wound Care (New Rochelle) 4(9): 560–82. doi: 10.1089/wound.2015.0635
Gottrup F, Jørgensen B, Karlsmark T, et al (2007) Less pain with Biatain-Ibu: initial findings from a randomised, controlled, double-blind clinical investigation on painful venous leg ulcers. Int Wound J 4(Suppl 1): 24–34. doi: 10.1111/j.1742-481X.2007.00312.x
Güiza-Argüello VR, Solarte-David VA, Pinzón-Mora AV, et al (2022) Current advances in the development of hydrogel-based wound dressings for diabetic foot ulcer treatment. Polymers (Basel) 14(14): 2764. doi: 10.3390/polym14142764
ISO (2003) Clinical investigation of medical devices for human subjects (ISO/DIS 14155-1:2003, part 1 and 2).
Lee EJ, Huh BK, Kim SN, et al (2017) Application of materials as medical devices with localized drug delivery capabilities for enhanced wound repair. Prog Mater Sci 89: 392–410. doi: 10.1016/j.pmatsci.2017.06.003/wound.2021.0016
Lindholm E, Löndahl M, Fagher K, et al (2019) Strong association between vibration perception thresholds at low frequencies (4 and 8 Hz), neuropathic symptoms and diabetic foot ulcers. PLoS One 14(2):e0212921. doi:10.1371/journal.pone.0212921
McDermott K, Fang M, Boulton AJM, et al (2023) Etiology, epidemiology, and disparities in the burden of diabetic foot ulcers. Diabetes Care. 46(1): 209–21. doi: 10.2337/dci22-0043
Nguyen HM, Ngoc Le TT, Nguyen AT, et al (2023) Biomedical materials for wound dressing: recent advances and applications. RSC Adv 13(8): 5509–28. doi: 10.1039/d2ra07673j
Niță O, Arhire LI, Mihalache L, et al. Evaluating classification systems of diabetic foot ulcer severity: a 12-year retrospective study on factors impacting survival. Healthcare (Basel) 11(14): 2077. doi: 10.3390/healthcare11142077
Norman G, Shi C, Goh EL, et al (2022) Negative pressure wound therapy for surgical wounds healing by primary closure. Cochrane Database Syst Rev 4(4): CD009261. doi: 10.1002/14651858.CD009261.pub7
Oliveira GM, Gomes Filho AO, Silva JGMD, et al (2023) Bacterial cellulose biomaterials for the treatment of lower limb ulcers. Rev Col Bras Cir 50: e20233536. doi: 10.1590/0100-6991e-20233536-en
Palao i Domenech R, Romanelli M, Tsiftsis DD, et al (2008) Effect of an ibuprofen-releasing foam dressing on wound pain: a real-life RCT. J Wound Care 17(8): 342–8. doi: 10.12968/jowc.2008.17.8.30797
Price P, Fogh K, Glynn C, et al (2007) Why combine a foam dressing with ibuprofen for wound pain and moist wound healing? Int Wound J 4(Suppl 1): 1–3. doi: 10.1111/j.1742-481X.2007.00310.x
Romanelli M, Dini V, Polignano R, et al (2009) Ibuprofen slow-release foam dressing reduces wound pain in painful exuding wounds: preliminary findings from an international real-life study. J Dermatolog Treat 20(1): 19–26. doi: 10.1080/09546630802178232
Saco M, Howe N, Nathoo R, Cherpelis B (2016) Comparing the efficacies of alginate, foam, hydrocolloid, hydrofiber, and hydrogel dressings in the management of diabetic foot ulcers and venous leg ulcers: a systematic review and meta-analysis examining how to dress for success. Dermatol Online J 22(8). doi: 10.5070/D3228032089
Salomé GM, Ferreira LM (2017) Impact of non-adherent Ibuprofen foam dressing in the lives of patients with venous ulcers. Rev Col Bras Cir 44(2): 116–24. doi: 10.1590/0100-69912017002002
Santema TB, Lenselink EA, Balm R, Ubbink DT (2016) Comparing the Meggitt-Wagner and the University of Texas wound classification systems for diabetic foot ulcers: inter-observer analyses. Int Wound J 13(6): 1137–41. doi: 10.1111/iwj.12429
Singla S, Garg R, Kumar A, Gill C (2014) Efficacy of topical application of beta urogastrone (recombinant human epidermal growth factor) in Wagner's Grade 1 and 2 diabetic foot ulcers: Comparative analysis of 50 patients. J Nat Sci Biol Med 5(2): 273–7. doi: 10.4103/0976-9668.136160
Tai FWD, McAlindon ME (2021) Non-steroidal anti-inflammatory drugs and the gastrointestinal tract. Clin Med (Lond) 21(2): 131–4. doi: 10.7861/clinmed.2021-0039
Wang X, Yuan CX, Xu B, Yu Z (2022) Diabetic foot ulcers: Classification, risk factors and management. World J Diabetes 13(12): 1049–65. doi: 10.4239/wjd.v13.i12.1049
