Clinical practice recommendations for diabetic foot attack: ‘This is how we do it’. Guidelines from the Latin American Diabetic Foot Association (ALAPID)
Received:
Published:
Authors: Martínez De Jesús FR, Carro GV, Cendejas Alatorre R et al
Citation:
Martínez De Jesús FR, Carro GV, Cendejas Alatorre R et al (2025) Clinical Practice Recommendations for Diabetic Foot Attack: This is How We Do It: Guidelines from the Latin American Diabetic Foot Association (ALAPID). Global Wound Care Journal 1(2 Suppl. 1): S1–S54.
Corresponding author:
Carlos Alberto Carrillo Bravo, Unidad Médica Nueva Clínica del Country, Bogotá, Colombia. Email. ccarrillo@praxisph.com.co
The aim of this report is to merge global and regional knowledge of the Saint Elian Wound Score System (SEWSS) into a document of clinical practice recommendations for diabetic foot syndrome. Under the concept, ‘This is how we do it’, the guidelines encompass definitions, classification, diagnosis, treatment, prognosis and prevention, aiming to reduce amputations and mortality. Originating from its local use in Mexico, the SEWSS has gained significant acceptance in Latin America and globally, particularly after its incorporation into the International Diabetes Federation’s Clinical Practice Recommendations for the Diabetic Foot in 2017. The SEWSS includes a triage system to assess the severity of the five types of diabetic foot attack (DFA) caused by ischaemia, infection, oedema, neuropathy (Charcot) or a combination. Patients with diabetes can progress from a low-risk stage to a foot attack, potentially resulting in major amputation or death. This review describes the progressive stages (1–3) of diabetic foot attack. The clinical details provided by the assessment of the 10 Saint Elian factors allow for a rational therapeutic approach, with an emphasis on prevention and medical treatment rather than wound care alone. Surgical treatment includes damage control for compartment syndrome and necrotising fasciitis secondary to a mixed attack from infection, oedema and acute Charcot. Recovery of pulsatile perfusion is achieved through angioplasty or bypass. These recommendations and guidelines, adopted by the Latin American Diabetic Foot Association, form the foundation of the SEWSS.
Introduction
Latin American culture has a set of traditions, beliefs and interpersonal relationships with a complex vision of the world, as well as ideologies, myths, legends and religious beliefs, with diverse backgrounds brought from the five continents of the world with Hispanic predominance. Consequently, a complex cultural and genetic diversity is created for its homogenisation, although with Hispanic and pre-Columbian cultural bases in the Mesoamerican, Inca roots and all their genetic diversity, mixed after the European conquest and the cultural genetic fusion between the white European, native indigenous and black African cultures.
Latin American culture not only suffers from problematised cultural, political and economic issues, but also from its own idealism due to its social inequalities. Pre-Columbian and African elements coexist in Latin American culture that do not respond to the western hegemonic tradition of modern European life. In this context, the presentation of a complication of diabetes, such as the diabetic foot (DF), presents not only the characteristics common to the development of this pathology anywhere in the world, but also those inherent to each region. In the case of Latin America, this includes the living conditions of people and their access to health, the elements available for diagnosis and treatment, the number of multidisciplinary teams dedicated to this pathology, and the beliefs of patients and their way of managing. This complication before the medical consultation, added to the time that elapses until the correct diagnosis and treatment by the health team, gives the DF its own characteristics that generate the need for management that is often different from that of the guidelines and recommendations of other regions. Diverse types of lesions of variable severity, bacteria with different sensitivity to antibiotics, variable degrees of ischaemia and late presentations are seen.
The low possibilities of prosthetic equipment after amputation in certain regions, and the high morbidity and mortality that patients have after amputation make DF salvage a fundamental procedure to avoid dependence and patient disability. This document arises from the need to adapt the recommendations to the “real world” in Latin America, taking into account the sociocultural and economic level of the region, and the availability of resources for the diagnosis and treatment of this pathology, with the aim of achieving the best possible results to keep patients on their feet.
The authors of these recommendations are members of the Latin American Diabetic Foot Association (ALAPID) and part of multidisciplinary teams dedicated to the care of patients with DF problems across various Latin American countries. These guidelines are based on the latest available literature on the subject, and the authors’ extensive experience in diagnosing and treating this complication. Major amputations are shown in cases of severe deformities and foot destruction. The Saint Elian Wound Score System (SEWSS) offers a platform for follow-up and comprehensive care.
Analysis of regional needs
Latin American culture encompasses a collection of traditions, beliefs and interpersonal relationships with a complex worldview, as well as ideologies, myths, legends and religious beliefs, with diverse origins brought from the five continents of the world, with a predominant Hispanic influence. This creates a complex cultural and genetic diversity that resists homogenisation. However, it retains Hispanic and pre-Columbian roots, particularly in Mesoamerican, Incan and other genetic diversities, which merged following the European conquest, and the genetic–cultural fusion between European white, native indigenous, and African black cultures.
Latin American culture not only grapples with the challenging array of cultural, political, economic and ideological variations, stemming not just from social inequalities, but also due to the coexistence of pre-Columbian and African elements that diverge from the dominant western tradition of modern European life.
The presence of indigenous elements is more pronounced in cultures with long pre-Columbian heritage (i.e. Mexico, Bolivia and Peru), while the Caribbean is African-descendant, and others, such as Argentina and Uruguay, exhibit more racially homogeneous characteristics. Within this history, beliefs and daily activities, such as gastronomy and nutrition, intricately linked to maize consumption, continue to endure.
Diabetes in Latin America
Currently, it is estimated that 15–20% of individuals with diabetes will develop ulcers over the course of their disease, with 30% of them facing amputations. A genuine issue is that 80% of major amputations in diabetes patients are avoidable (International Diabetes Federation, 2017).
Multidisciplinary management allows proper treatment, including revascularisation and specialised care, which, in many cases, can save limbs. Within 5 years, 50% of people with diabetes who undergo an amputation lose the other extremity (Buggy and Moore, 2017; Cassidy et al, 2021; Meloni et al, 2021. Globally, a major diabetes-related amputation occurs every 30 seconds (International Diabetes Federation, 2017). Mortality following amputation reaches 13% to 40% in the first year, 35% to 65% in the third, and 39% to 80% in the fifth year. (Pinilla et al, 2013).
The percentage of new foot ulcer cases correlates with diabetes duration, age, male sex, smoking habits and the level of preventive measures in place. Prevention is the only way to decrease economic and social impact, needing multidisciplinary teams to reduce amputation rates. It is crucial to implement programmes in public and private institutions with necessary equipment and well-trained personnel. Prompt initiation of technical and clinical activities upon the arrival of a diabetes patient can prevent or at least delay catastrophic complications.
The DF has an annual incidence of 2%, which occurs in 19–34% of patients with diabetes throughout their lives, according to European studies (IWGDF, 2019; Oe et al 2025). In Latin America, the DF is the reason for hospitalisation in 3.7% of cases and in 20% of hospitalised patients with diabetes.
The global and Latin American impact of the SEWSS
The Latin American SEWSS has been widely disseminated and referenced in multiple impactful publications, book chapters, renowned award conferences and clinical guidelines, including its significant global contribution in the International Diabetes Federation Clinical Practice Recommendations of the Diabetic Foot 2017 guidelines (Martinez-De Jesús 2010a, 2010b; Martínez-De Jesús et al, 2012a, 2012b, 2012c, 2012d, 2017; Consejo de Salubridad General, 2013; Guías de práctica clínica, 2017, Sistema de San Elián en las Guías de Orientación técnica, 2018.
Noteworthy conferences highlighting the system’s contribution in various regions include:
- The Multidisciplinary International Limb and Amputations prevention Conference. Classificazioni del piede diabético: la classificazione di San Elian e migliori di altre, Milan, Italy, February 2013.
- The Saint Elian comprehensive surgical approach for diabetic foot wounds. Diabetic Foot International Summer School of Surgery. Bergamo, Italy, 2017.
- Certification for Physicians to Prevent Diabetic Foot Syndrome: The Saint Elian System. International Diabetes Federation Congress. Abu Dhabi, United Arab Emirates, 2017.
- The Saint Elian score for the diabetic foot syndrome. A dynamic and detailed system to reduce lower-extremity amputation. The foot attack in LATAM. The International Diabetes Federation Global Congress. Busan, Korea, 2019.
Regarding the regional dissemination of the SEWSS for the Americas, its contribution stands out in various publications as part of clinical practice guidelines from medical associations and health ministries of Chile, El Salvador, Paraguay and Mexico (Consejo de Salubridad General, 2013; Mexican Association of General Surgery, 2014; Certification for Physicians, 2017; Guías de práctica clínica, 2017; Sistema de San Elián en las Guías de Orientación técnica, 2018; Ministry of Health, Salvador, 2021; Salvadoran Social Security Institute-, 2022). Its inclusion in the classification chapter of various guidelines converges on the work that representative delegates from the Americas finalised in the document Declaration of the Second Summit of the Americas for Diabetic Foot (Martinez-De Jesús et al, 2017). The systematised model integrates all preventive and therapeutic interventions based on severity grades and types of foot attack, including the therapeutic model for correcting aggravating factors or attack type and treatment by healing phases.
In conclusion, the SEWSS provides a validated tool with global impact and wide application and dissemination, primarily in Latin America (Soares et al, 2015; Huang et al, 2017; Maslak et al, 2017; Niță et al, 2021; Monteiro- Carro et al, 2022; Carro et al, 2023a, 2023b). Its intended legacy is to empower healthcare professionals in the treatment and prevention of diabetes complications, particularly the DF, mainly through strict glucose control. The system motivates the creation of multidisciplinary groups to prioritise prevention and, based on that, provide prompt treatment to improve therapeutic outcomes and reduce amputations. The DF attack (DFA), according to the SEWSS, presents five modalities corresponding to the aggravating factors of Saint Elian or their combined presentation. The system contributes regionally and globally to the definition, classification, diagnosis, prognosis, treatment, prevention and triage for acute DFA.
The Diabetic Foot Attack
The concept of the DF does not merely refer to the feet of individuals with diabetes. The DFA describes a devastating and catastrophic syndrome associated with necrotising fasciitis and compartment syndrome, presenting significant challenges for medical and surgical decision-making. DF syndrome can manifest as either an acute or chronic attack, characterised by one or more wounds with varying aetiology, complexity and severity. These wounds result from diverse types of attacks caused by infection, ischaemia, oedema and neuropathy (Charcot), often associated with compartment syndrome and/or necrotising fasciitis. This increases the risk of amputation and/or death in individuals with diabetes.
This syndrome can evolve suddenly into a severe and acute DFA, characterised by acute inflammation, rapid and progressive skin and tissue necrosis, and a systemic response. It can be chaotic and unpredictable, with the potential for limb loss and high mortality, necessitating urgent intervention (Vas et al, 2018; Carro et al, 2020). Under these terms, we reasoned that necrotising fasciitis, compartment syndrome and DFA are synonymous with similar clinical presentations, differing only in their severity. DFA should be considered a medical emergency, requiring prompt recognition, urgent and immediate treatment, and/or referral to a specialised multidisciplinary team to ensure the survival of both the limb and the patient. A DFA can rapidly escalate over hours to limb- and life-threatening proportions (Sistema de San Elián en las Guías de Orientación técnica, 2018). The SEWSS is an important tool for the DF triage in acute DFA, and enables wounds to be classified according to the urgency and severity of the lesion (Martínez-De Jesús, 2010b).
Types and stages of acute DFA
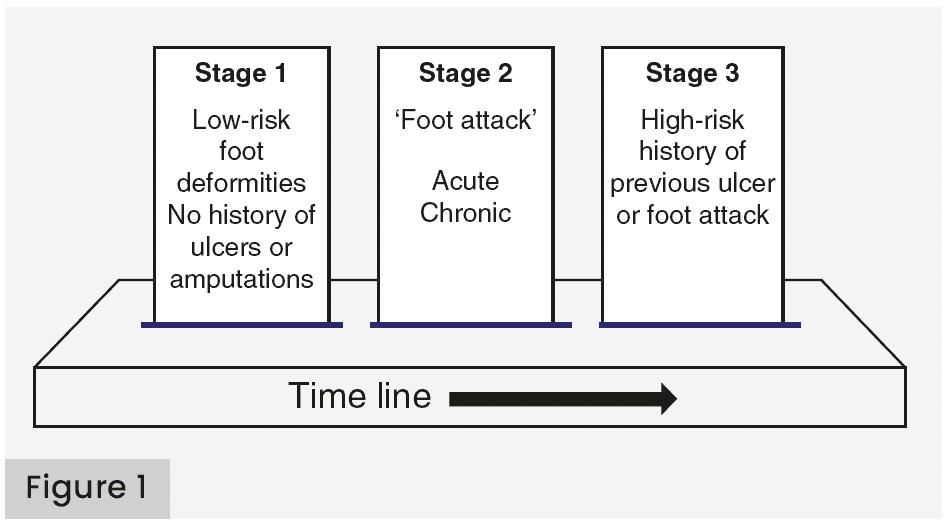
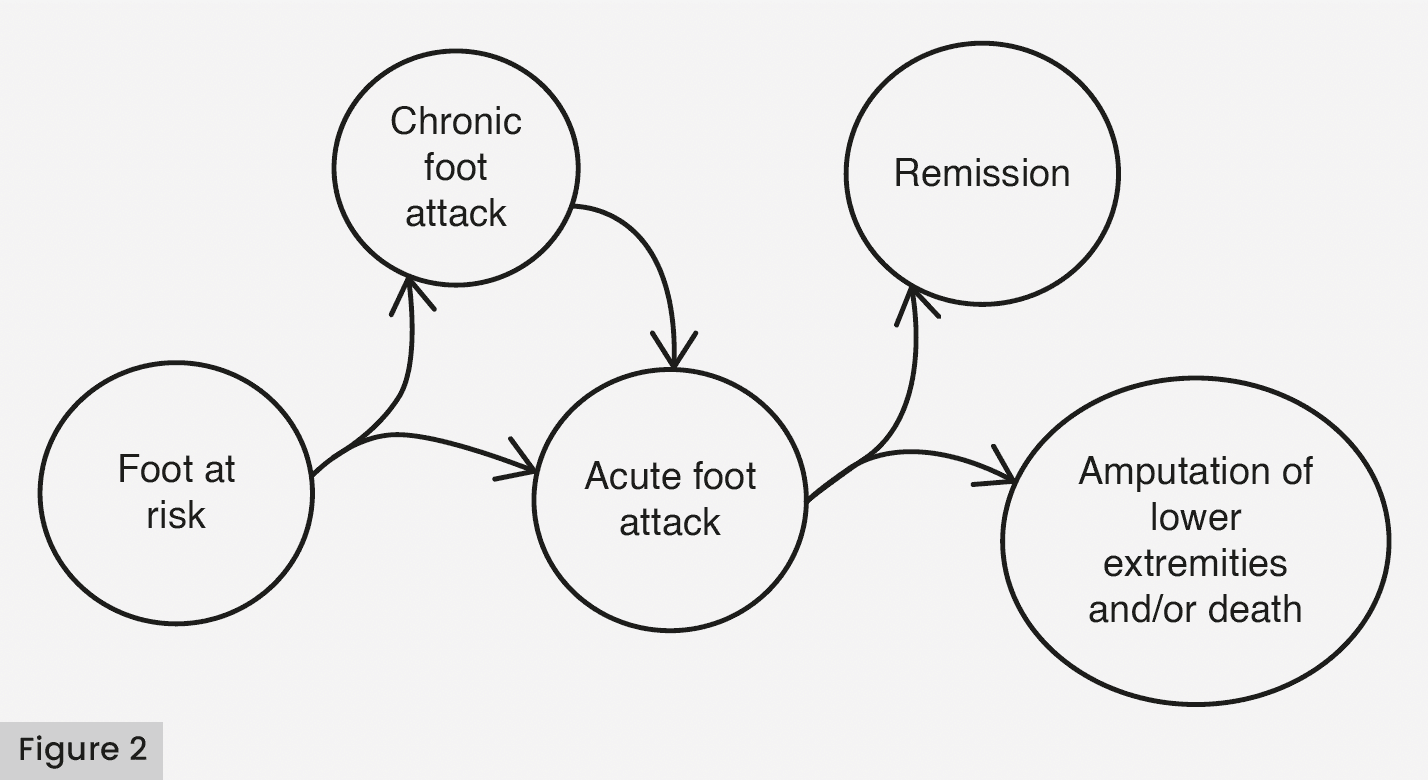
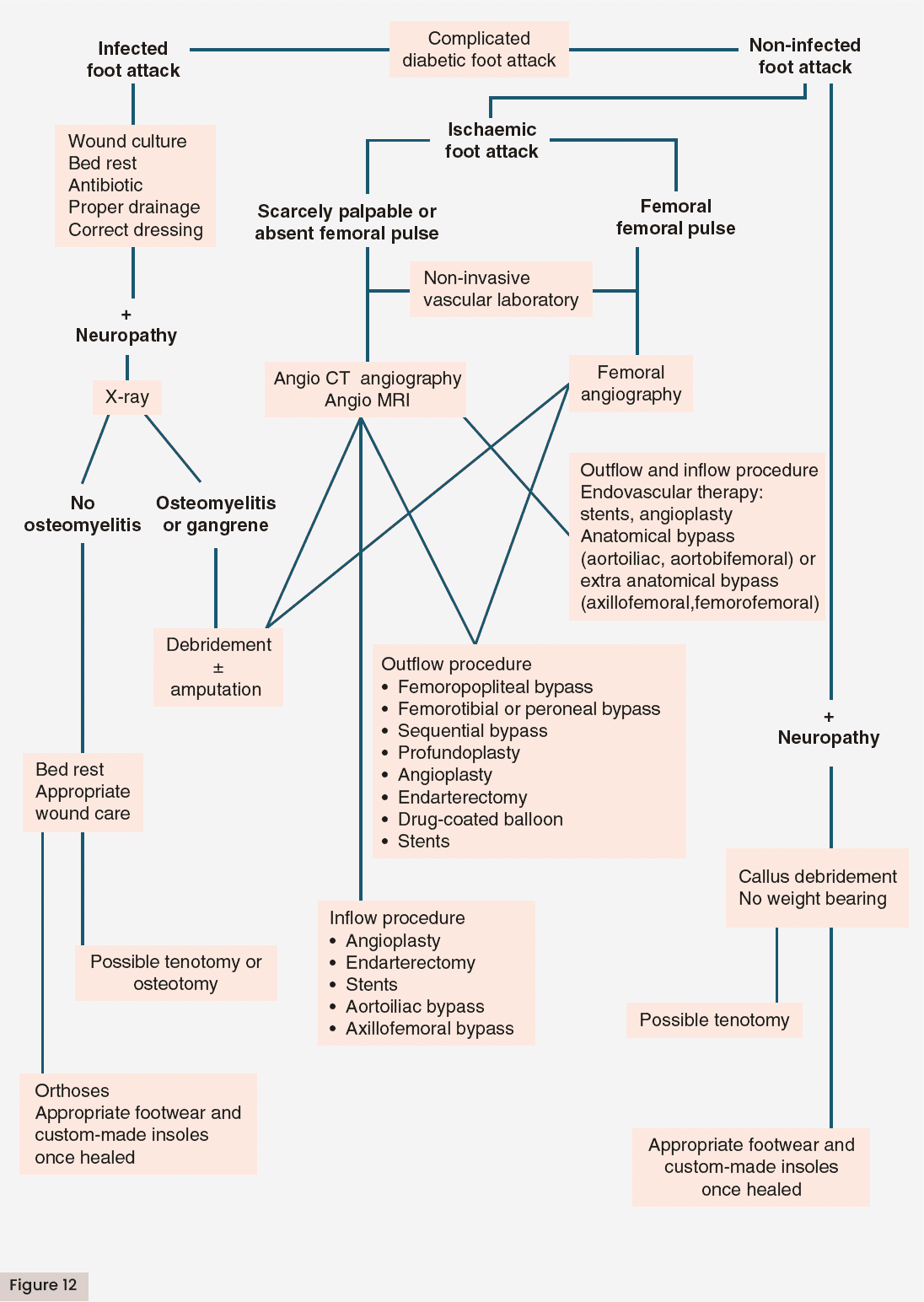
Patients with diabetes can progress from a low-risk stage to a foot attack, potentially resulting in major amputation or death [Figure 1]. This document describes the DFA progressive stages (1–3) that imply an acute and severe presentation, which threatens the affected limb and even the life of the patient. It is a term that arises from the need to ease the identification of those patients who require urgent intervention, giving it a sense of urgency and seriousness, reinforcing the concept that ‘time is tissue’. Stage 3 is posterior to the DFA once remission is achieved, or an adverse consequence of major amputation or death [Figures 1 and 2].
Types of DFA
Five situations have been identified in which urgent intervention is needed:
- The infected DFA: the foot presents with a serious infection and rapid progression of necrosis that requires hospitalisation, rapid debridement of devitalised tissue, drainage of collections and antibiotic treatment.
- Ischaemic DFA: implies progressive and evolving ischaemia, and requires urgent revascularisation.
- Neuropathic DFA: Charcot neuroarthropathy in the acute phase that requires rapid identification and discharge.
- DFA: due to oedema that originates from compartment syndrome that causes fluid retention in the interstitial space produced by various underlying diseases. This must be diagnosed and treated to eliminate this morbidity and to be able to improve and treat this ulceration.
- Mixed DFA: the combination of two other types.
Timely recognition of these presentations is essential to start adequate treatment and improve patient outcomes.
The concept of the DFA has emerged among those who care for patients with this condition. This term is used to refer to DF which, due to its serious condition and imminent worsening, has a poor prognosis if not diagnosed, and treated promptly and adequately, emulating the concept of cerebrovascular attack and heart attack, used to denote the seriousness of these entities and the urgency with which they must be treated.
The term DFA has been used since at least 2013 in the UK. This was later emphasised by Vas et al (2018), who described its forms of presentation and defined the concept of ‘golden hours’, referring to the importance of the first hours in the opportune treatment of the DFA. This term confers a powerful sense of urgency, both for physicians and for other members of the health team.
During the past few years, there have been two significant advances in the understanding of the DFA. First, it was understood that the person who suffers from this condition experiences multiple relapses and requires rapid access to a multidisciplinary team whenever necessary to provide proper treatment before the lesion progresses to necrosis or severe infection. Second, early diagnosis of ischaemia and urgent revascularisation are essential to improve the outcome and reduce amputations.
The different treatments required by a patient with DFA include hospitalisation, and immediate drainage of collections and debridement of sloughed tissue, offloading, intravenous application of broad-spectrum antibiotics prior to taking samples for cultures, and, in some cases, revascularisation. Failure to identify this condition and delay in the establishment of treatments seriously worsens the prognosis, and loss of the limb and an elevated risk of death can occur in a matter of hours or days. There are three types of DFA according to Vas et al (2018):
- Infected DF: foot with acute inflammation, erythema, collections, extensive devitalised tissue, moist necrosis, localised or entire limb oedema, with or without systemic signs of sepsis.
- Ischaemic DF with progressive ischaemia: these are generally patients with critical ischaemia who are already at the end of the evolution, with pain at rest, with or without tissue loss and who have progressed in recent days.
- Charcot neuroarthropathy (CN) in the acute phase: refers to those patients who present with an oedematous, erythematous foot, without pain (although this is not exclusive), who often consult the guard and are treated as cellulitis, but without indicating discharge. Often the correct diagnosis is not made, and they tend to present deformities that are irreversible.
These last two cases are considered by Vas et al (2018) as atypical or overlapping forms of DFA, but, in addition to that, two more types are described, such as the DFA due to oedema and the mixed DFA described by Martinez-De Jesus et al (2021) as part of the Latin American SEWSS. The triage of DFA includes five types of foot attack: ischaemic, infectious, oedema, neuropathic and mixed.
Stages of the DFA
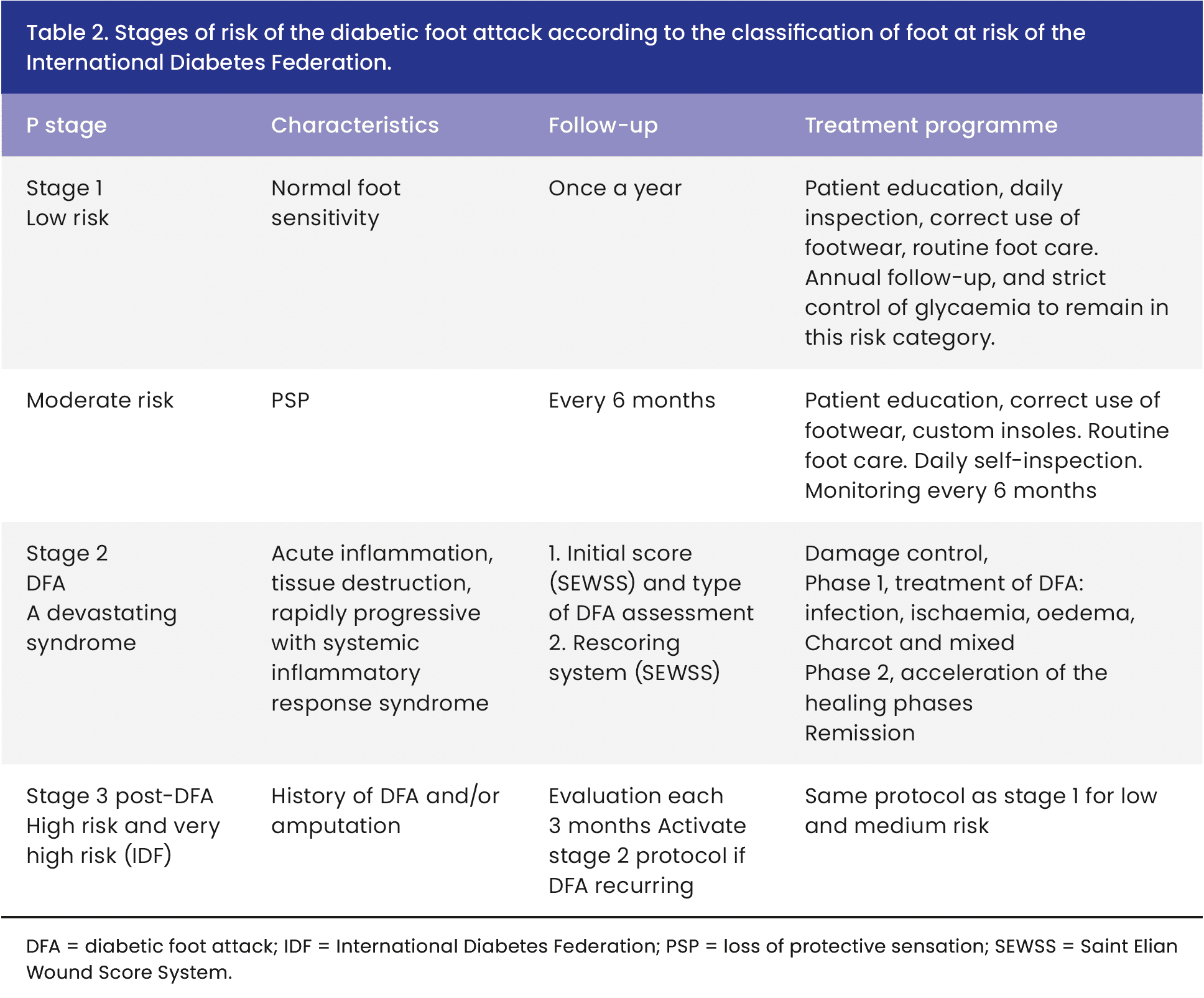
People with diabetes can progress from a low-risk foot stage to a foot attack that will remit or, in contrast, progress to amputation and/or death. Figures 1 and 2 show the following evolutionary stages. Stage 1: low-risk foot. They are people with diabetes with no history of foot ulceration or foot attack at the time of their visit. Stage 2: the DFA. It can remit with successful healing with or without minor amputations or, on the contrary, progress to a major amputation or death. Stage 3: high risk. It is the latter state, with a history of a healed wound with minor amputations or major amputation due to DFA with an extremely elevated risk of limb loss or developing a new DFA. Figure 2 describes the scheme that allows us to describe the risk stages, characteristics, follow-up and therapeutic plan, which is detailed in Tables 1, 2 and 3.
The Latin American Saint Elian Wound Score System
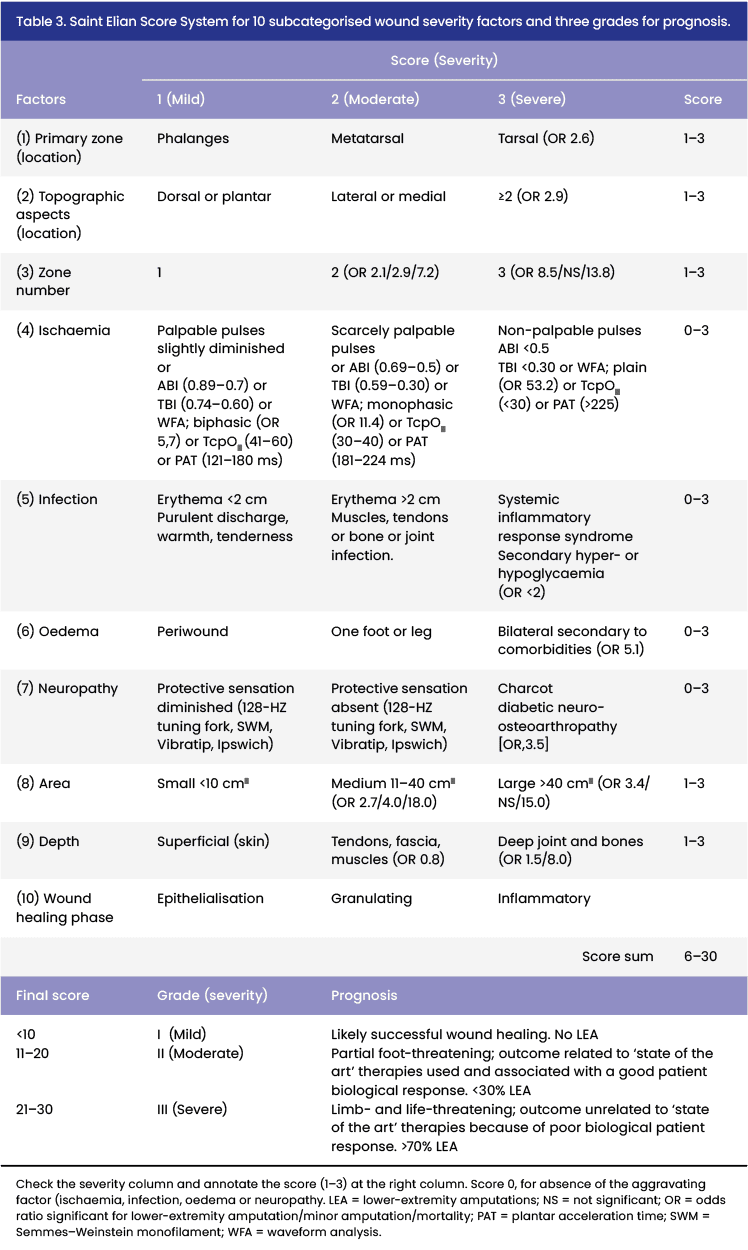
The San Elian structure and function consist of 10 factors: three for anatomical location, four for aggravating and three for tissue destruction. Anatomical factors encompass initial zone, anatomical aspects and number of areas. Aggravating factors include ischaemia, infection, oedema and neuropathy, while tissue destruction involves area, depth and healing phases (Martínez-De Jesús, 2010b).
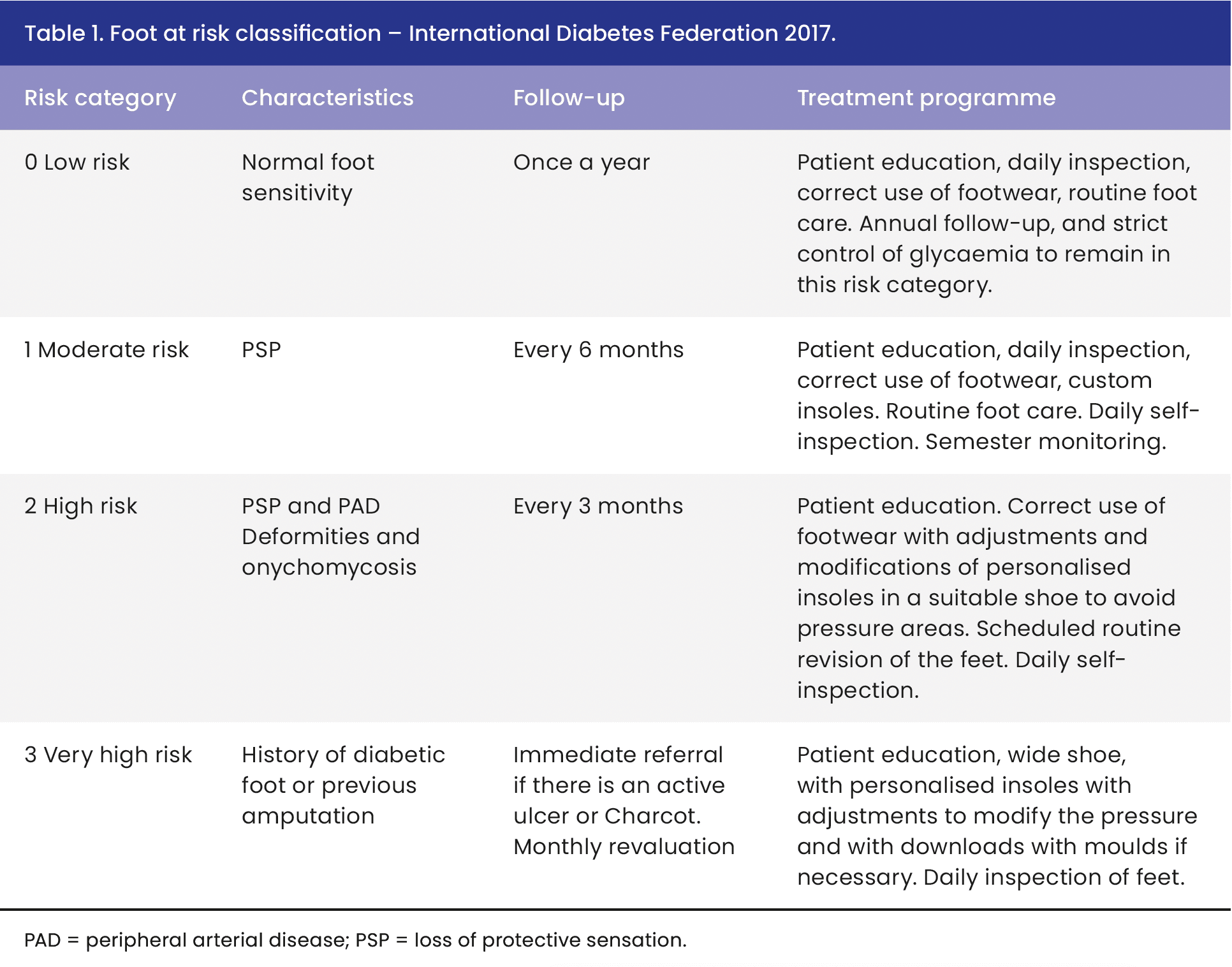
A checklist system for scoring and grading the severity of DFA was published by the International Diabetes Federation, utilising the 10 SEWSS variables, which determine prognosis into three levels, indicating the risk of amputation or death [Figure 3]. The SEWSS is a globally impactful tool used in Latin America for broad application. It is crucial to ensure uniformity in the observation of variables, as different medical professionals may treat this complication differently. Training in diabetes complication treatment and prevention, especially DF care, is paramount, along with strict glucose control to prevent this devastating complication.
May these efforts and others contribute to the prevention of diabetes complications, particularly the most devastating complications. Patients with robust prevention efforts and multidisciplinary care teams should not suffer from such complications (Martinez-De Jesús, 2010a, 2010b; Martínez-De Jesús et al, 2012a; 2012b, 2012c; Sistema de San Elián en las Guías de Orientación técnica, 2018).
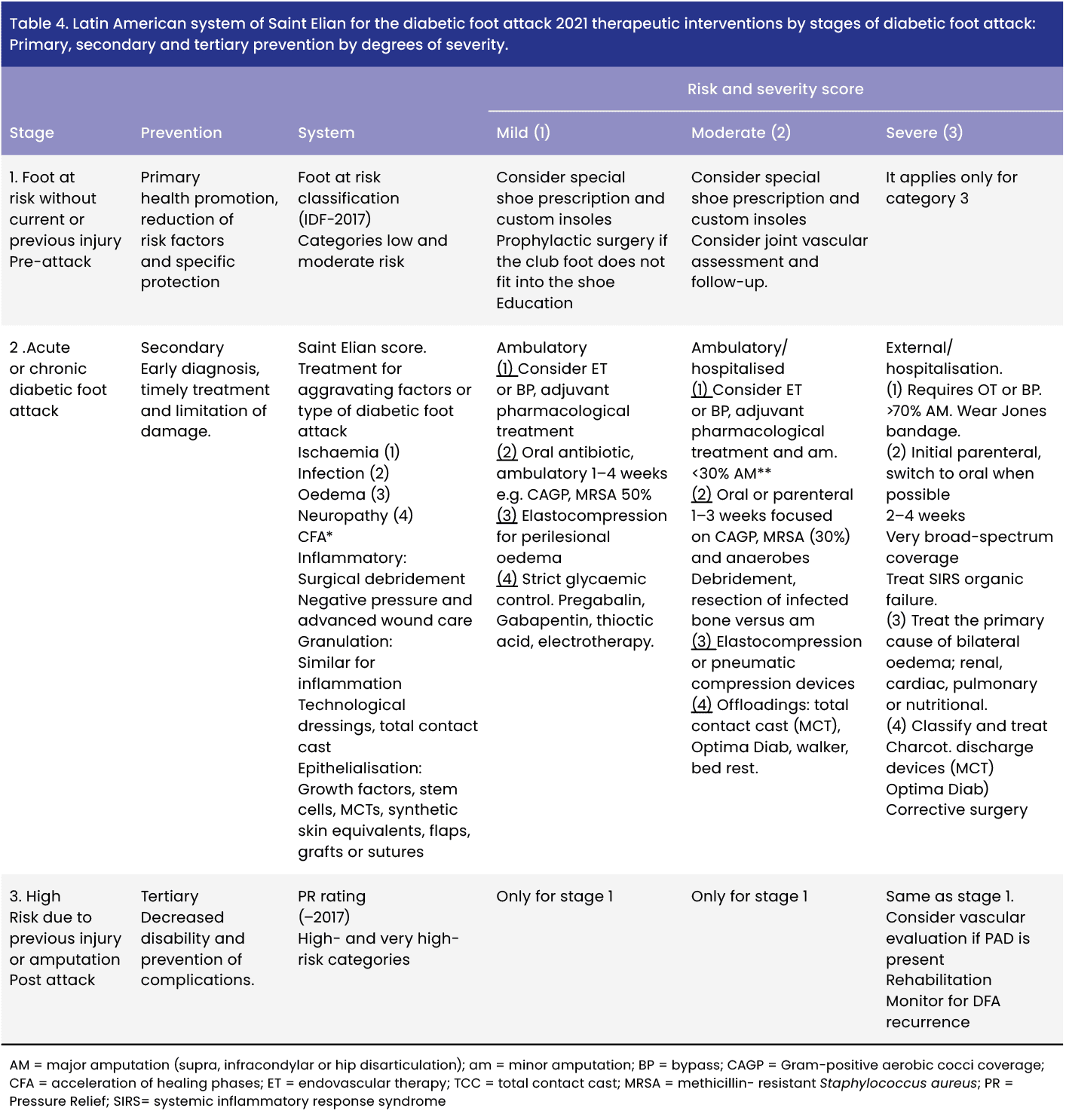
The SEWSS has been subjected to methodological scrutiny under a rigorous scientific process of internal and external validation that allows a model with a comprehensive care approach to achieve the aforementioned purposes (Soares et al, 2015; Huang et al, 2017; Maslak et al, 2017; Niță et al, 2021; Monteiro-Carro et al, 2022; Carro et al. 2023a, 2023b). The clinical importance of the SEWSS for the DF is related to its relevance for diagnosis, prognosis, treatment, monitoring of the progress of DFA and prevention. The usefulness and application of the system in each of these functions is described below [Table 4].
Initial assessment and follow-up of the DFA
A typical DFA may begin as a simple injury, but could rapidly escalate over hours to limb-threatening proportions. This may be a completely new presentation or a sudden deterioration in the history of a known neuropathic ulcer, in limb ischaemia, or both, where delays in recognition or intervention pose a significant risk of lower-extremity amputation.
The first evaluation of a DFA can be done in an outpatient setting in an emergency room or in a hospital setting. When evaluating a patient with a suspected DF infection, a detailed history and physical examination should be performed to decide the most appropriate intervention.
Diagnosis and score of wound factors
The diagnosis and score are based on a correctly performed medical history, which includes anamnesis and physical examination directed to a detailed DF evaluation, focusing on arterial, neuropathy and ulcer characteristics, avoiding omissions. The severity grading includes the diagnosis of the severity score of 10 factors [Table 3]:
- First location.
- Topographic aspects.
- Number of affected areas.
- Ischaemia.
- Infection.
- Oedema.
- Neuropathy.
- Depth.
- Area.
- Healing phase.
These are subcategorised with an ascending severity score from mild (1 point) to severe (3 points).
The assessment sequence includes: to diagnose the initial location with anamnesis about where the wound begun; the foot aspects affected by the attack must be submitted to a careful inspection to score their severity; how many zones are involved; the presence and severity of ischaemia of the involved leg; the presence of infection and its severity; the presence and severity of oedema; the severity of neuropathy and extent of the damage of the Charcot foot; the wound depth with prone to bone test; X-ray film or MRI; wound extent by simple rule measures; and by inspection and monitoring the wound healing.
Assessment of the topographic wound variables: location, foot aspect and number of affected zones
The primary location is determined by directly questioning the patient regarding the place where the lesion began. A primary localisation in the digital area is mild, a metatarsal is moderate and a tarsal is classified as severe. Plantar or dorsal aspects are classified as mild, moderate (if medial or lateral aspects are involved) and severe (if all aspects are involved). One affected area is considered mild, two as moderate and involvement of the entire foot as severe (multiple lesions >2). Neuropathic ulcers often develop on the plantar surface of the foot or in areas of bony prominence. Ischaemic and neuroischaemic ulcers most commonly develop at the toes or the lateral edges of the foot.
Assessment of aggravating factors
3.1.1.2.1. Technical details of specific tests for ischaemia
3.1.1.2.1.1. Non-invasive vascular tests for diagnosis
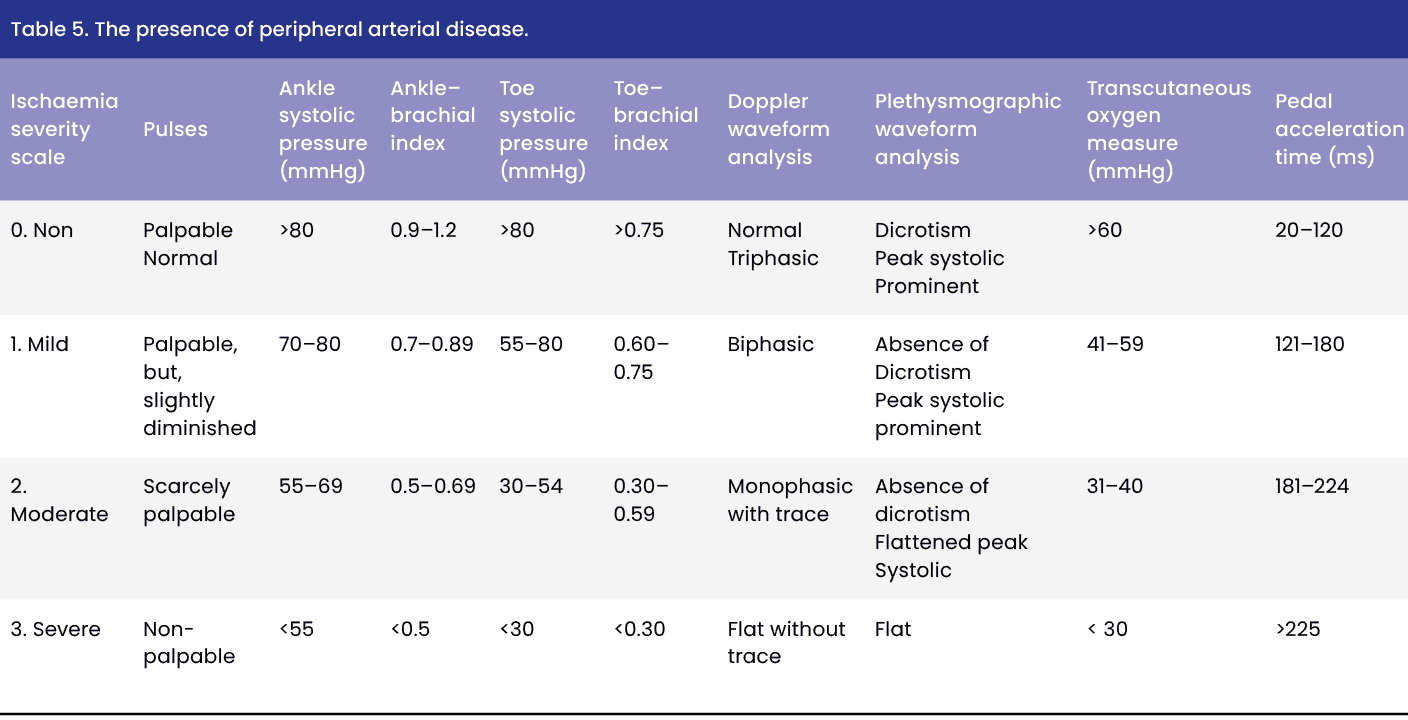
The palpation of a strong pulse, a triphasic waveform of the pedal artery, and measuring the ankle pressure and ankle brachial index (ABI) using a Doppler instrument are used. The presence of an ABI of 0.9–1.3 or a triphasic wave in the foot pulse excludes the presence of peripheral arterial disease, as well as a toe–brachial index ≥0.75 [Table 5].
Pulse palpation
In patients who have an ischaemic DFA, pulses palpation must be strictly performed: femoral, popliteal, anterior tibial or pedal and posterior tibial in their first assessment; and in each re-classification of the SEWSS, tibial pulses should be reassessed. In patients with diabetes, especially those in remission or at elevated risk for foot complications, pulse palpation should be performed in each consultation without exception. The pedal pulse is palpable on the dorsum of the foot, lateral to the flexor tendon of the first toe; in 10% of cases, this pulse is not palpable. The posterior tibial pulse is palpable 2 cm posterior to the medial malleolus or midway between the medial malleolus and the Achilles tendon. The popliteal pulse is palpated with the knee flexed to 30° with both hands, and is found slightly lateral to the midline of the popliteal fossa. The femoral pulse is palpable in the inguinal region in the middle of an imaginary line from the anterior superior iliac spine to the pubis. For palpation of the dorsal or posterior tibial arteries foot pulses, strong tibial pulses are classified as (0, non-ischaemic); palpable, but slightly diminished (1, mild); threadlike and barely palpable (2, moderate); and non-palpable (3, severe) (Martinez-De Jesús et al, 2022).
Ankle systolic pressure
Systolic pressure of tibial arteries is assessed using a sphygmomanometer and an arterial flow Doppler; the systolic pressure of the pedial and posterior tibial artery are measured (the one with the highest range is taken as the reference), with normal being >80 mmHg. In some cases, when there is calcification of the middle layer (Mönckeberg’s calcinosis), there may be no compressibility of the arteries, and pressures at this level cannot be evaluated. The ankle pressure and ABI may be falsely elevated due to calcification of the pedal artery. In selected cases, other tests (e.g. measurement of the great toe pressure or transcutaneous oxygen pressure [TcpO2]) are useful to assess the vascular status of the foot. The ABI and the systolic pressure of the tibial arteries should be performed in all patients.
ABI
The ABI is the result of the ratio between the highest value obtained in the measurement of the posterior tibial arteries and the pedia between the highest value of the systolic pressure of the arm. The ABI can be used in diabetes patients, but the values must be interpreted with caution and according to the clinical situation, since they may be normal or falsely elevated in patients with Mönckeberg calcification. The normal result for a patient with diabetes is 0.91–1.3. An ABI <0.9 shows peripheral arterial disease due to obstruction secondary to atherosclerosis. An ABI >1.3 is suggestive of the presence of a calcification process of the arterial media layer, these patients may present peripheral arterial disease between 58% and 84% due to decreased elasticity as a result of arterial wall rigidity, but other measures are needed. Studies, such as the toe–brachial index or plethysmography, should be performed to irrefutably verify peripheral arterial disease.
Toe systolic pressure
A toe systolic pressure reading should be performed in patients with tissue loss and absence of pulses. It requires a special finger cuff and sphygmomanometer, a flow Doppler or a plethysmographic waveform Doppler.
It should be performed on the first or second finger. Its normal value is 80 mmHg [Figure 4].
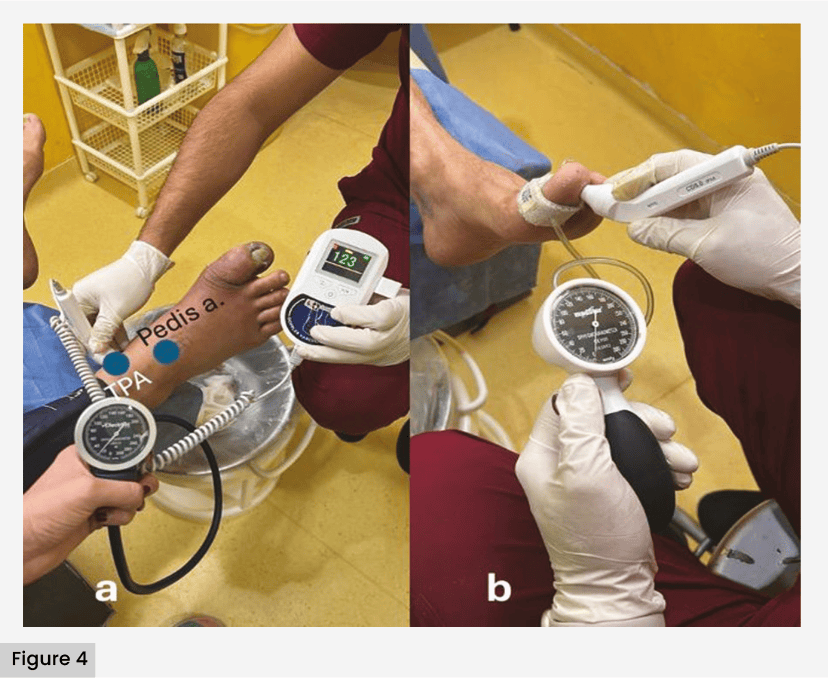
Toe–brachial index
The toe–brachial index consists of dividing the systolic pressure of the finger by the systolic pressure of the humeral artery; the result must be >0.75.
Pulse volume plethysmography
Pulse volume plethysmography measures pulse volume variations in the limb. It is useful when the arteries are rigid and non-compressible in diabetes patients; it also makes it possible to study the vasomotor response ability, after the provocation of reactive hyperaemia.
Flow Doppler
Flow Doppler assesses the morphology of the wave, which can be triphasic (flow and elasticity of the normal wall), biphasic or monophasic (abnormal), which will indicate obstruction, stiffness of the arterial wall or even endothelial dysfunction (monophasic laminar or pandiastolic flow).
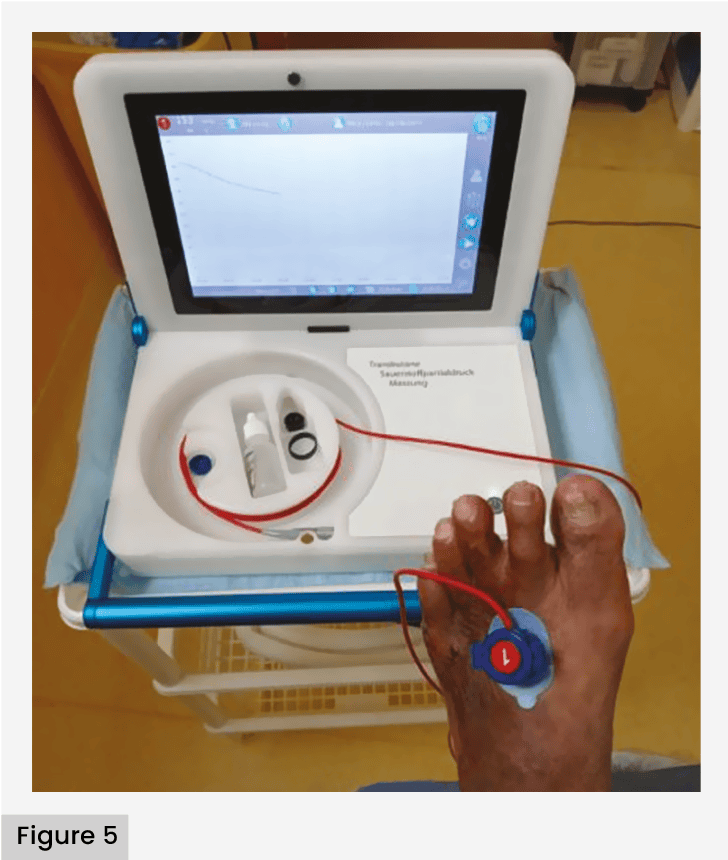
TcpO2
TcpO2 performs the perfusion measurement at the microvascular level of the foot; preferably at the level of the angiosome that is involved in the wound; the normal value is 70 mmHg, and the minimum value suggested for the success of an adequate epithelialisation of the wound is 30 mmHg [Figure 5]. If the resource is available, it is recommended to perform the oxygen challenge test to assess other pathologies as the origin of hypoperfusion at the foot level (Congestive Heart Failure, chronic obstructive pulmonary disease, oedema) or assess the benefit of management with a hyperbaric chamber. In patients with obstructive ischaemia (atheromatous obstructive arterial disease), there will be no response to 100% oxygen placement with a mask, and in the event of presenting an increase in the curve and the level of oxygen tension at >100 mmHg, the origin of hypoperfusion will be a problem of O2 diffusion (congestive heart failure, chronic obstructive pulmonary disease, anaemia), and in these cases the use of hyperbaric therapy is recommended. Haemodynamic parameters, such as ABI and systolic pressures of tibial arteries, can be falsely elevated due to calcinosis of the media layer in diabetes patients and patients with end-stage renal disease. For these reasons, the combination of all studies is necessary. In one study, it was shown that systolic pressure failed to identify 42% of patients with critical limb-threatening ischaemia, and finger pressure measurement and TCpO2 are more accurate in predicting the risk of amputation at 1 year (pedal pressure <30 mmHg, TcpO2 <10 mmHg).
The results of studies that define critical limb-threatening ischaemia and the need for revascularisation are flat wave morphology, ankle systolic pressure <50 mmHg, ABI <0.5, toe systolic pressure <30 mmHg, Toe-Brachial Index <30 mmHg and TcpO2 <30 mmHg. Duplex–Doppler ultrasound is recommended as the first option to assess arterial anatomy, when grade 3 (severe) of the SEWSS ischaemia classification is reached based on hemodynamic and perfusion criteria (Norgren et al, 2007; Met et al, 2009; Høyer et al, 2013; Conte et al, 2019; Teso et al, 2021).
Duplex–Doppler ultrasound
The sensitivity of the duplex–Doppler ultrasound to detect critical stenosis with haemodynamic changes that are classified as ≥50% stenosis varies from 89% in the iliac segment, 68% in the femoropopliteal segment and 82–90% in the iliac segment. It is also useful in assessing hibernating vessels distal to the obstruction that cannot be assessed on angiography.
Plantar acceleration time
When performing duplex–Doppler ultrasound, it is recommended to perform plantar acceleration time, which consists of assessing the velocity of the systolic peak at the level
of the arteries of the foot and, thus, confirming the parameters of critical ischaemia in the foot. After revascularisation, it is recommended to perform plantar acceleration time immediately or early after surgery to assess the success of the procedure. It has been shown that measuring the acceleration time towards the systolic peak is superior in predicting the decrease in diameter and flow compared with the measurement of velocity, and provides real-time physiological information on flow haemodynamics at the arterial level of the foot. It can be measured with any duplex–Doppler ultrasound with a 10–15-MHz linear transducer of the vessels of the foot (arcuate, 1st metatarsal, medial plantar, lateral plantar). Its normal value is 20–120 ms (Teso et al, 2021).
3.1.1.2.1.2. Invasive vascular tests
Contrast studies are recommended to assess urgent revascularisation in patients with ischaemic DFA with any foot lesion and where the non-invasive vascular laboratory results are systolic pressure of tibial arteries <50 mmHg, ABI <0.5, toe–brachial index <0.3, toe systolic pressure <30 mmHg and TcpO2 <30 mmHg (American Diabetes Association, 2021). Contrast studies are not recommended as the first option to assess arterial obstructive disease in patients who have not been diagnosed with critical limb-threatening ischaemia by non-invasive methods.
Invasive vascular studies are recommended for patients with an ischaemic DFA with a wound, regardless of the results of non-invasive studies, if the wound does not show improvement (there is no decrease in the SEWSS) after 4–6 weeks of treatment using the best available therapy.
- Angio-tomography: useful for patients with aortoiliac disease and obesity, and for patients who want to assess restenosis of stents, fractures or thrombi. Its use is not recommended for evaluation below the knee due to the presence of calcification that can be confused with iodinated contrast.
- Resonance angiography: presents less confusion between contrast and calcium than CT angiography, and gadolinium is less nephrotoxic than iodinated contrast.
- Digital subtraction angiography: considered the gold standard in imaging of the arterial tree; especially useful in assessing the arteries below the knee, and the anatomy of the plantar and pedal arteries. It can be performed with CO2 contrast medium in patients with kidney damage, and it is proposed to be performed exclusively when endovascular revascularisation is planned at the same time as the study.
3.1.1.2.2. Technical details of specific tests for assessment of neuropathy
For the diagnosis of diabetic neuropathy in its first stages, it is essential to enquire about the type of pain experienced by our patients, the intensity and frequency of the pain. By using the Michigan Neuropathy Screening Instrument and tabulating the data, we can differentiate it from vascular pain and other types of pain that may occur in diabetes patients.
However, during the DFA physical examination of the foot, the assessment should be done using tools, such as a 128-Hz tuning fork, reflex hammer and a 10 g monofilament (American Diabetes Association, 2021).
a) The 10 g monofilament (Semmes–Weinstein 5.07 [detects loss of protective sensation]).
- First apply the monofilament to the patient’s hands (or to the elbow or forehead) to show them what it feels like to feel it.
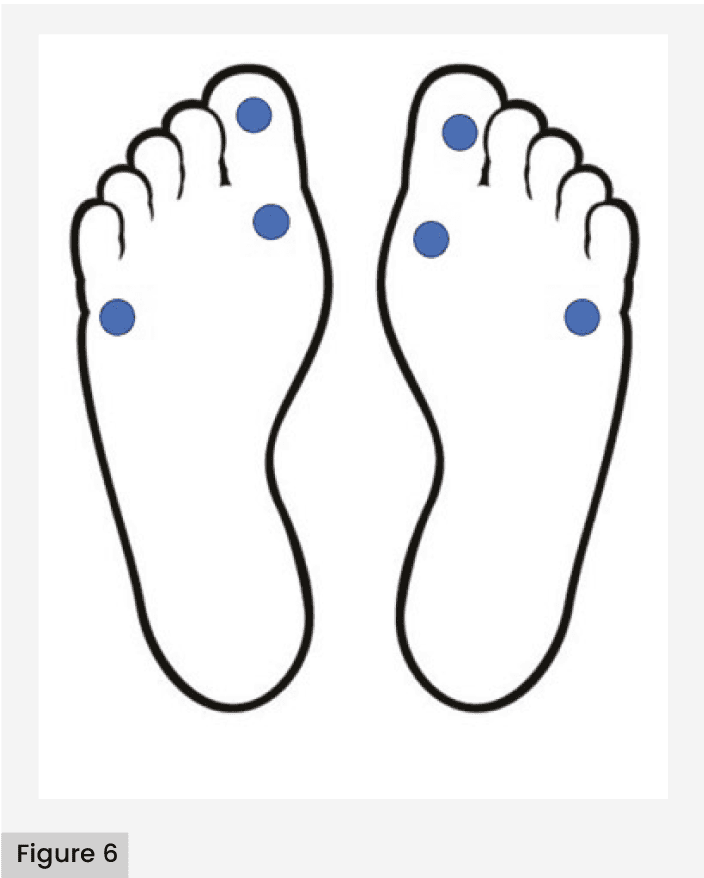
- Assess three different points on both feet, selecting from the points shown in Figure 6.
- Make sure the patient cannot see if the examiner is applying the monofilament.
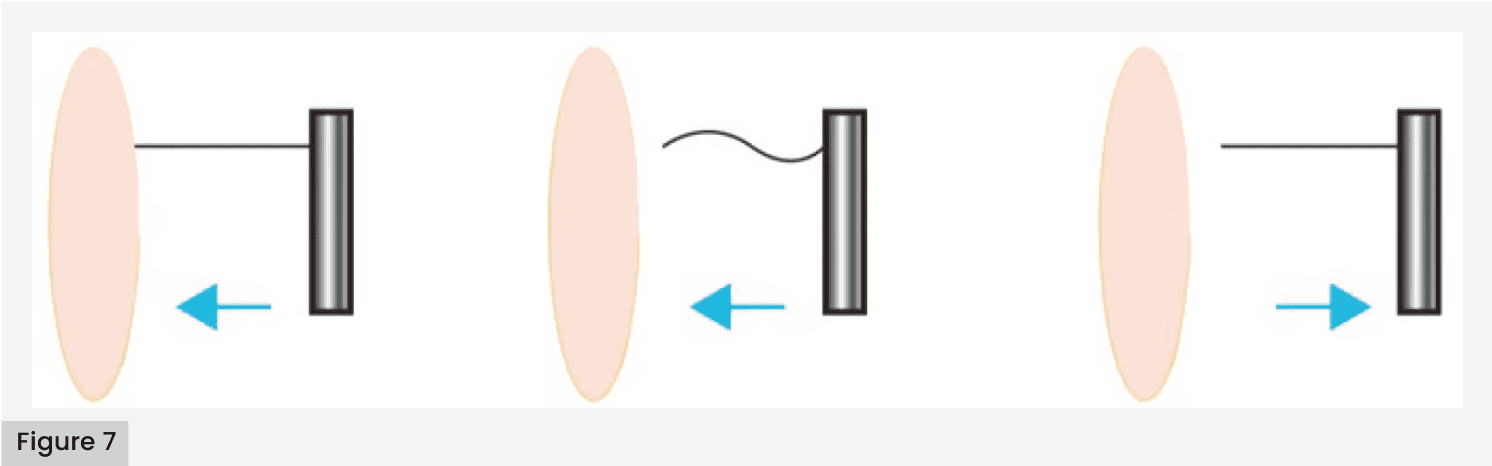
- Apply the monofilament perpendicular to the skin surface [Figure 7] with enough force to cause the filament to bend or loop.
- The total duration of the application (contact with the skin) and removal of the filament should last approximately 2 seconds.
- Do not apply the filament directly to an ulcer, callus, scar or necrotic tissue.
- Do not allow the filament to slide across the skin or create repetitive contact at the test point.
- Press the monofilament onto the skin and ask the patient if they feel the applied pressure (yes/no) and then where they feel the pressure (e.g. left sole/right heel).
- Repeat this application twice on the same point, but alternating with at least one dummy application, in which the filament is not applied (a total of three questions per point).
- Protective sensitivity is present at each point if the patient answers correctly in two of the three applications, and absent with two of three incorrect answers.
- Encourage patients during the evaluation with positive comments.
- Monofilaments tend to lose deformability temporarily after being used multiple times in the same day, or permanently after prolonged use. Depending on the type of monofilament, we suggest not using the monofilament after 24 hours after testing 10–15 patients, and replacing it after using it on 70–90 patients.
b) Tuning fork (128 Hz, detects the loss of vibratory sensitivity).
- First, apply the tuning fork to the patient’s wrist (or elbow or clavicle) to show them what it feels like to feel it.
- Make sure the patient cannot see if the examiner is applying the tuning fork.
- Apply the tuning fork [Figure 8] to a bony part on the back of the distal phalanx of the first finger (or on another finger if the hallux is absent).
- Apply the tuning fork perpendicularly, with constant pressure.
- Repeat this application twice, but alternate with at least one simulated application in which the tuning fork does not vibrate.
- The test is positive if the patient answers correctly to at least two of the three applications, and negative if two of the three answers are incorrect.
- If the patient cannot feel vibrations in one finger, repeat the more proximal test (e.g. malleolus, tibial tuberosity).
- Encourage the patient during the evaluation with positive comments.
c) The Ipswich touch test, also called the Ipswich tactile test (detects loss of protective sensation, when a 10 g monofilament or 128 Hz tuning fork is not available). The test is in reasonable agreement with these tests for deciding the presence of protective sensation, but its accuracy in predicting foot ulcers has not been established.
- Explain the procedure and make sure everything is understood.
- Instruct the patient to close their eyes and say ‘yes’ when touched.
- The examiner touches the tip of the patient’s index finger lightly and sequentially on the balls of the first, third and fifth toes of both feet for 1–2 seconds
- When touching, do not push, hit or poke.
- Protective sensation is likely when tactile sensation is not detected at two or more sites.
3.1.1.2.3. Technical details of specific tests for assessment of infection
Clinical data are collected and scored according to the SEWSS scale for infection severity [Table 3] (Martínez-De Jesús, 2010b).
Infection in the foot of a person with diabetes represents a serious threat to the foot and the affected lower limb, which is why it must be evaluated and treated quickly. Since all ulcers are colonised with potential pathogens, infection should be diagnosed by the presence of at least two signs or symptoms of inflammation (redness, warmth, induration, pain/tenderness) or the presence of purulent discharge. Unfortunately, these signs may be diminished in the presence of neuropathy or ischaemia, and systemic findings (e.g. pain, fever, leucocytosis) are often absent in cases of mild and moderate infection. Infections should be classified using the Infectious Diseases Society of America classification included as part of the assessment of the SEWSS [Table 3] as mild (superficial with minimal cellulitis), moderate (deep or extensive) or severe (accompanied by systemic signs of sepsis), as well as whether there is the presence or not osteomyelitis.
The infectious DFA shows clinical modalities of varying severity degrees, as follows: as an Infected ulcer; osteomyelitis as part of the DFA; osteomyelitis as part of a mixed foot attack in Charcot foot; and as part of compartment syndrome secondary to a mixed attack, including, Charcot, oedema and infectious necrotising fasciitis.
3.1.1.2.3.1. Diagnostic test and clinical modalities of osteomyelitis
Another factor to take into account is the presence of osteomyelitis, which can be present in a moderate or severe infection and must be thoroughly investigated; sometimes, it is possible to observe the exposure and bone destruction, or it can be suspected in the presence of a ‘sausage toe’ with sinus tract, purulent discharge and drainage of bone fragments that suggests osteomyelitis (Martinez-De Jesús et al. 2012; Lipsky et al, 2020; American Diabetes Association, 2021). A simple and practical method for its diagnosis is the probe-to-bone test, which is performed by exploring the ulcer using a metallic probe and testing if it touches bone (positive probe-to-bone test). This method has a high sensitivity and specificity for osteomyelitis (0.87 and 0.91, respectively). In the presence of an infected in DFA, radiographs provide limited value for diagnosing osteomyelitis or determining the timing of surgery. Surgical intervention should not be delayed due to the absence of radiographic findings, because cortical bone changes typically appear late and radiographs lack the sensitivity needed to reliably exclude osteomyelitis (Lipsky et al, 2020; Carro et al 2023).
However, simple radiography of the foot can help to obtain more information on the condition of the deep tissues, looking for the presence of gas or detecting foreign bodies. MRI is the study that provides the highest diagnostic accuracy for osteomyelitis (sensitivity 90% and specificity 79%), and it could also help locate collections in deep tissues; however, its use is limited, and it does not constitute a first-line study.
It is important to take samples during debridement, and more so if bone curettage is performed, since this will allow a definitive diagnosis of osteomyelitis by histopathological study and to obtain the causative agent when culturing the sample. When the patient presents with an infected DFA accompanied by a systemic inflammatory response (presence of ≥2 of the following criteria: temperature >38ºC or <36ºC, leukocytes >12,000 or <4,000 or bandaemia >10%, heart rate >90 BPM , respiratory rate >20 breaths per minute, PaCO2 <32 mmHg, hyperglycaemia or severe hypoglycaemia secondary to sepsis), it should be considered as a serious infection (SEWSS score of 3 points; Martinez-De Jesús et al, 2012; Lipsky et al, 2020; Carro et al, 2023).
Evaluate patients with a DF infection for osteomyelitis, especially those with long-standing, deep ulcers, or ulcers located directly over a bony prominence. Examine the ulcer to determine if bone tissue can be seen or palpated with a sterile, metallic instrument. In addition to clinical evaluation, consider taking plain radiographic images in most patients, looking for evidence of osteomyelitis, soft tissue gas or foreign bodies.
DF osteomyelitis poses significant challenges in clinical management due to its varied presentations and associated complications. Each presentation of DF osteomyelitis requires specific diagnostic and therapeutic approaches to optimise patient outcomes. There are different clinical presentations of DF osteomyelitis and their respective management strategies.
- a) Osteomyelitis as part of DFA
Is an acute, severe infection presenting with rapid tissue necrosis and abscess formation. The diagnosis includes clinical features, probe-to-bone test and radiographs. Radiographs may not always show osteomyelitis early due to the rapid progression of the infection. - b) Sausage toe
Consists of a swollen, erythematous toe with oedema and an ulcer, typically affecting the phalanges. Clinical features, probe-to-bone test and radiographs are key for the diagnosis. The treatment includes a 6–8-week course of antibiotics, with a high success rate without the need for amputation. Bone biopsy or MRI is not required unless there is diagnostic uncertainty or failure of initial treatment. - c) Osteomyelitis superimposed on Charcot foot
It comprises a chronic ulceration in a deformed foot, often involving the midfoot or hindfoot. For the diagnosis, MRI is required for accurate assessment due to similar radiographic appearances to Charcot’s changes. Additional diagnostic tools include laboratory tests and sometimes fludeoxyglucose PET scans. The treatment includes complex surgical interventions to stabilise the foot and prevent recurrent ulcers. Surgery may include realignment osteotomy, fusion or other procedures to correct deformity and improve foot stability. - d) Chronic osteomyelitis with small ulceration
Chronic indolent ulcers over bony prominences, often secondary to previous unsuccessful surgeries or persistent sinus tracts. Clinical features, radiographs and laboratory tests are required for diagnosis. Antibiotic therapy guided by surgical or transcutaneous biopsy and surgery are often required to remove infected bone, with minor amputations or osteotomies being common.
For clinically infected ulcers, a tissue sample should be obtained for culture (if Gram-stained culture is available); avoid obtaining ulcer samples for culture by means of smears. The causative pathogens of foot infection (and their antibiotic susceptibility) vary according to geographic, demographic and clinical situations, but Staphylococcus aureus (alone or with other organisms) is the predominant pathogen in most cases. Chronic and more severe infections are often polymicrobial, with aerobic Gram-negative and anaerobic bacilli accompanying Gram-positive cocci, especially in warmer climates.
3.1.1.2.3. Technical details of specific tests for assessment of oedema
Oedema is a sign that appears in many diseases, and manifests as soft tissue swelling due to fluid accumulation in the interstitial compartment. It arises if an imbalance occurs between the forces that regulate the passage of liquid from one compartment to another. If the passage of water is abundant from the intravascular to the interstitial compartment, oedema appears. This fluid extravasation can respond to physical or chemical factors, from an increase in intravascular pressure in the return circulation, to an increase in the permeability of the vascular wall or a decrease in the levels of molecules that maintain intravascular fluid (proteins). Oedema can be noticed locally, as it occurs in the lower extremities or as generalised swelling (hands, abdomen). Oedema occurs in patients with diabetes in cases of diabetic nephropathy, congestive heart failure, venous insufficiency, liver failure or cirrhosis and lymphoedema.
Diabetic nephropathy is a microvascular complication of diabetes secondary to poor metabolic control and aggravated by the presence of arterial hypertension. Hyperglycaemia causes an increase in intracellular glucose, resulting in the activation of alternative metabolic pathways, such as the polyol pathway, with the decisive participation of enzymatic elements, such as aldose reductase, generating kidney damage and proteinuria. Functional and structural changes occur at the renal level, such as glomerular hypertrophy and mesangial proliferation, which lead to a modification of corpuscular haemodynamics, and the stimulation of cell proliferation and hypertrophy processes.
The most frequent symptoms are peripheral oedema, bipalpebral oedema and involuntary weight gain due to fluid retention, which leads to an increased pressure in the soft tissues on the vascular supply, causing a compartmental syndrome that leads to the formation of ulcers secondary to this oedema. This oedema causes or favours the breakdown of the skin and the subsequent appearance of an ulcer, and this ulcer tends to become infected by bacteria, such as methicillin-resistant S. aureus, Streptococcus and Escherichia coli, among others.
The accumulation of liquid is more evident in the regions of the body where loose tissue abounds, such as the ankles, the eyelids and the sacral area. The skin over the swollen area may be stretched or shiny. If pressed, it leaves a mark or pit that slowly disappears. Oedemas may have a local (ankle or leg), regional (whole extremity) or general distribution [Table 3]. Inflammatory oedemas, in contrast to the others, are painful, hot and reddish. The most common symptoms are swelling, stretched or shiny skin, pitting and a feeling of heaviness.
To diagnose oedema, the clinical interview is of utmost importance, since it attempts to determine the clinical profile of this fluid retention or the origin of the oedema. A simple and pathognomonic sign is the Godet’s sign (pitting oedema), a print under the digital pressure over the oedematous skin.
For better patient care and for adequate symptom control, we must carry out a personalised follow-up to assess the efficacy of the indicated therapeutic measures. Complete physical examination is a fundamental pillar, together with assessment of the body’s fluid and solid components using bioimpedance.
We must perform simple analytical studies that include the assessment of renal function and excretion of water/electrolytes, cardiovascular overload, and protein metabolism.
Likewise, a multidisciplinary team made up of vascular surgery, endocrinology, gastroenterology, and rehabilitation and physiotherapy.
3.1.1.3. Assessment of tissue destruction wound factors
Determining the depth of foot ulcers can be difficult, especially in the presence of callus or necrotic tissue. To aid ulcer evaluation, any neuropathic or neuroischaemic ulcer that is surrounded by callus or necrotic soft tissue should be debrided initially or as soon as possible. However, an uninfected ulcer that shows signs of severe ischaemia should not be debrided.
3.1.2. Score and classification for prognosis
The SEWSS consists of 10 variables specific to the injury, which in turn are subdivided into mild, moderate and severe, to finally obtain a score that categorises into: grade 1 (mild, good prognosis for successful healing), grade 2 (moderate, partial threat, the results are dependent on adequate therapy applied [lex artis medica ad hoc] and associated with the patient’s biological response) and grade 3 (serious, threatening to the affected limb and life; Martinez-De Jesús, 2010b). The results are independent of the correct therapeutic and biological response of the patient. This system applied in the initial assessment and during the follow-up of the chronobiology of the patient’s lesion allows us to understand the effect of the ideal therapy on the evolution of the lesion [Table 3].
The DFA may be of a sudden onset without history of previous attack, or it may constitute the exacerbation of a chronic process due to ischaemia, neuropathy or both. Therefore, the delay in the diagnosis or early intervention generates a significant risk of major amputation.
3.1.3. SEWSS for the triage of DFA
The SEWSS provides a guide with the aim of prioritising the medical treatment of people with DF according to the probability of benefiting from such attention to survive or to limit the damage and function of the extremity [Table 6].

DFA is an emergency and should be treated as such, with the same importance as a head injury or any other emergency procedure that we know can cause death if not resolved.
At presentation, there is an acutely swollen foot with rapidly progressive necrosis to the skin and tissues. There are significant systemic symptoms of sepsis or systemic inflammatory response that can lead to major amputation of the limb and even death.
We currently have a measurement, classification, diagnosis and prognosis system – the SEWSS – which includes anatomical factors, aggravating factors and tissue affection. This score allows us to assess patients in a timely manner and, thus, be able to offer treatment immediately and urgently in the case of any type of DFA [Figures 2 and 3].
The DFA and the triage for the DFA is a new concept that is consolidated among those who care for patients with DF, and arises to identify those who represent a significantly higher risk of limb loss and who, with adequate, timely and correct attention, can improve the prognosis. This concept imposes urgency for the identification of the case and rapid decision-making to avoid the most serious complication, which is amputation. Five types of DFA have been identified, as named before, whose initial treatment can change the prognosis, although in practice, there are cases in which ischaemia and infection are combined (mixed DFA), and treatment of both is required at the same time. These cases are potentially the most serious. Identifying these patients is the first step to improving their care, although other measures need to be adopted.
3.1.4. SEWSS for monitoring progress of the DFA
The severity score and prognostic grading are assessed using a checklist format of the ten factors as many times as necessary (Martínez-De Jesús et al, 2012). The frequency can vary from daily, every 3 days or weekly reassessment. Changes in the score allow for continuous monitoring to implement proactive therapeutic actions [Figure 9].

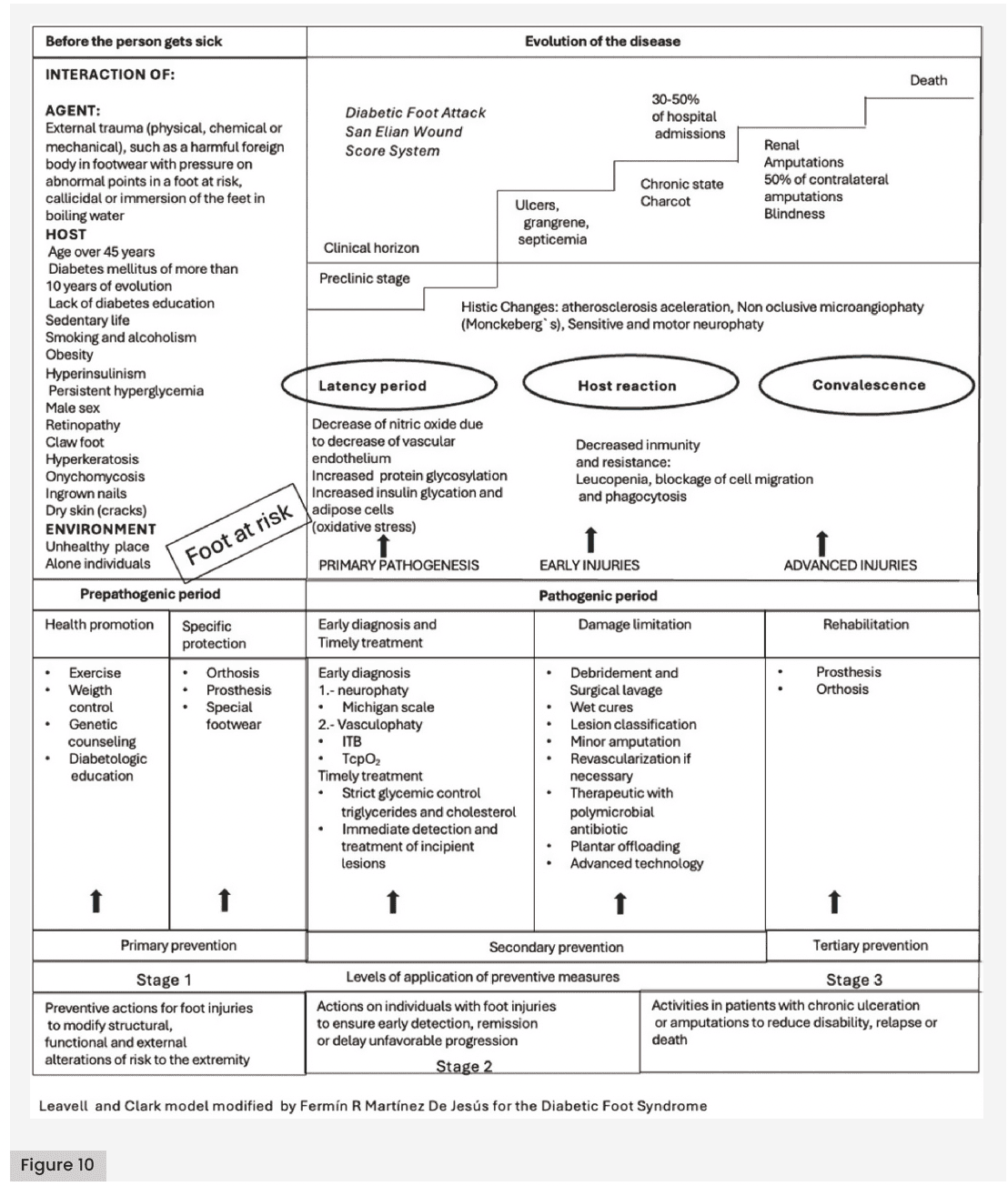
3.1.5. Prevention
The system provides a platform for primary, secondary and tertiary prevention according to the Leavell and Clark model [Figure 10], and the risk classification of the International Diabetes Federation for the DF (International Diabetes Federation, 2017). The preventive model includes guidelines for referral and counter-referral based on different levels of care. The potential value of the system lies in the inclusion of demographic parameters in a detailed and unified scoring system, facilitating communication and collaboration among colleagues worldwide to reduce amputations in diabetes patients. Individuals with diabetes can progress from a low-risk foot stage to a foot attack that may regress or, conversely, evolve into amputation and/or death [Figure 10] (Mani, 2010; Martinez-De Jesús et al, 2022).
3.2. Treatment of the DFA according to the SEWSS
3.2.1. Treatment for aggravating factors or type of DFA
The treatment is effective regardless of the multiple commercial therapeutic modalities if it focuses primarily on the correction of the four aggravating factors (ischaemia, infection, oedema and neuropathy) according to the severity score (0–3) and, subsequently, the acceleration of the healing phases [Figures 9, 10 and 11].
Martínez-De Jesús (2010) has highlighted the relevance of 10 wound-related factors as part of a dynamic system that interact throughout the course of a DFA, influencing the outcome either positively or negatively. The four aggravating factors are correlated with the type of DFA. Multivariate analysis of these ten variables indicates that DFA secondary to ischaemia is the primary contributor to major amputations. Furthermore, univariate analysis of the ischaemia factor revealed a significant odds ratio for major amputations across all severity grades (from mild to severe), along with other SEWSS factors associated with tissue destruction in terms of depth and extent.
The absence of sensation (moderate neuropathy) and Charcot (severe) show an increased chance for limb loss compared with non-neuropathic foot attacks. The chance of major amputations is increased by bilateral severe oedema, usually secondary to renal failure coincident with infection and Charcot as part of a mixed attack with compartment syndrome and necrotising fasciitis. The appropriate aggressive surgical treatment and antibiotics increase the chance of wound healing success without lower legs extremities amputations.
The surgical treatment of the SEWSS to reduce lower-extremity amputations and mortality includes different procedures according to the objectives to solve the type of DFA [Table 7]. The surgery to contain the damage of compartment syndrome is performed to avoid local tissue destruction and systemic dissemination of the infection.
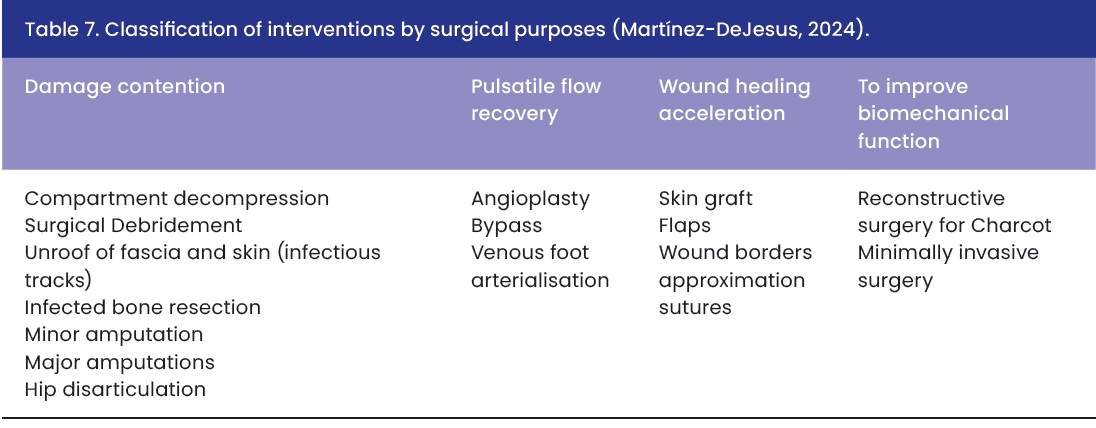
In case of DFA secondary to ischaemia, the goal is to recover pulsatile blood flow and pressure to the distal foot. At the third column of Table 7, the procedures to accelerate wound healing with surgical sutures, skin grafts and flaps are described. Finally, when the DFA is secondary to Charcot foot, the surgical treatment must be oriented to correct the foot deformities after remission of the acute attack. During acute Charcot foot attack, the conservative treatment is total offloading with total contact cast.
3.2.1.1. Treatment of the infected DFA
The surgical solution addresses the deadly triad of oedema, Charcot neuroarthropathy, and infection by targeting each component through timely intervention, aiming to reduce complications and improve patient outcomes
Once the presence of infection has been diagnosed, it is a priority to classify it to determine its severity according to the SEWSS assessment equivalent to the Infectious Diseases Society of America, which replaces other classifications, such as Wagner or the University of Texas, that are no longer used in various countries because of their limited low value to characterise infection. Infection treatment should be considered according to its severity. The Infectious Diseases Society of America defines a mild infection (SEWSS infection scoring 1 point) as the presence of infection limited to the skin or subcutaneous tissue, cellulitis <2 cm, induration, heat, pain and purulent discharge (Lipsky et al, 2020; Carro et al, 2023). In these cases, treatment should be with empirical antibiotic therapy, aimed at the use of monotherapy that covers the most frequent bacteria, Gram-positive aerobic cocci (dicloxacillin, amoxicillin/clavulanic acid, clindamycin, cephalosporins, quinolones).
Although the literature recommends use of monotherapy, another study carried out in Latin America recommends using a combination of two antibiotics, amoxicillin/clavulanic acid and trimethoprim/sulfamethoxazole, especially if osteomyelitis is suspected. Continuing antibiotics until wound closure is not justified, as this increases the likelihood of antimicrobial resistance and the cost of the treatment (Capobianco et al, 2010; Carro et al, 2023).
Cleaning of the wound, removal of helomas and debriding the wound, as well as unloading the affected area, should be carried out routinely. Management is ambulatory and does not require hospitalisation. It is essential to take care and control of the patient in the DF clinic, assessing the lesion every 2–3 days; once the wound is cleaned, interactive dressings can be used to accelerate its healing.
In wounds without clinical evidence of infection, antimicrobial therapy is not recommended. When the patient presents a moderate infection with a SEWSS score of 2, characterised by cellulitis >2 cm, and the presence of abscesses, necrosis, fasciitis, septic arthritis, or gangrene with or without osteomyelitis, the presence of a DFA due to infection should be potentially suspected, so initial hospital management should be considered, with the application of intravenous antibiotics, absolute rest and urgent debridement. Once the infection process is controlled, oral antibiotics can be continued on an outpatient basis (Lipsky et al, 2020). The selection of the empirical antibiotic is aimed at treating polymicrobial flora, with broad-spectrum drugs or combinations: cephalosporins, quinolones or amoxicillin/clavulanic acid, combined with clindamycin or ciprofloxacin, ticarcillin/clavulanic, piperacillin/tazobactam, linezolid alone or with aztreonam, ertapenem and, when osteomyelitis is suspected, the combination of amoxicillin/clavulanic acid and trimethoprim/sulfamethoxazole is suggested (ciprofloxacin is not recommended, as it does not have satisfactory coverage for osteomyelitis caused by S. aureus, due to resistance arising from prolonged treatment). Antibiotics should be administered for 2–4 weeks (Carro et al, 2022).

In the presence of an infected DFA, timely recognition is essential as soon as possible, without delaying surgical treatment, since this constitutes the cornerstone of the treatment; surgery should not be delayed waiting for the impregnation with antibiotics, keeping in mind that the risk of amputation increases by 1.6-fold per day of delayed surgery (Aragón-Sánchez et al, 2012). Urgent surgery is recommended in the presence of DFA due to infection, which includes infections characterised by gas in tissues, deep abscesses or necrotising fasciitis. The surgical procedure should aim to drain abscesses, eliminate all infected necrotic tissue, open all existing fistulous tracts and thoroughly irrigate the wound with warm saline solution. This procedure must be repeated as many times as necessary.
When systemic inflammatory response syndrome is diagnosed, infection must be scored as severe (SEWSS score, 3 points). At this stage, the possibility of loss of the limb and death increases from 18 points; therefore, timely treatment must be instituted as soon as possible, and close monitoring of these patients must be maintained (Huang et al, 2015; Carro et al, 2022, 2023). There could be no fever or leucocytosis, so a potentially serious deep tissue infection should not be ruled out in the absence of these data, and it is necessary to have a high index of clinical suspicion to perform early surgical interventions.
Patients with a SEWSS grade 2 classification >17 points or grade 3 should be strictly monitored in ambulatory care. Comorbidities or systemic inflammatory response syndrome require patient hospitalisation to start multidisciplinary management, recommending antibiotics administered intravenously, selecting empirically a combination of a double or triple scheme: quinolones with clindamycin, imipenem/cilastatin or vancomycin/ceftazidime with or without metronidazole; multidisciplinary management should focus on stabilisation, control of present comorbidities, and emergency surgical interventions to eradicate necrotic and infected tissue.
3.2.1.2. Necrotising fasciitis and compartment syndrome
Acute compartment syndrome occurs when there is increased pressure within a closed osteofascial compartment, resulting in impaired local circulation. Acute compartment syndrome is considered a surgical emergency, since, without proper treatment, it can lead to ischaemia and eventually necrosis of the affected foot.
Necrotising fasciitis is an uncommon, rapidly progressive and often aggressive bacterial infection that causes extensive necrosis or inflammatory destruction of the subcutaneous tissue, skin, muscle and fascia. Necrotising fasciitis is a feature of DFA secondary to infection. Rapid diagnosis of the disease is mandatory, as delays in initiating aggressive treatment negatively influence the outcome. Specific clinical signs may not always be present, making accurate and timely diagnosis difficult. Necrotising fasciitis is found in 79.4% of patients with foot attack, and with aggressive surgical therapy, an 85% wound healing success rate is achieved. The mortality rate is significant, at 9.2%, and major amputations occur at a rate of 1.9%, which can be minimised with appropriate surgical intervention (Iacopi et al, 2015; 2023).
Necrotising fasciitis presents as a surgical emergency. It is characterised by the rapid progression of inflammation, leading to extensive tissue necrosis and destruction. However, the diagnosis might be missed or delayed due to variable and non-specific clinical presentations, contributing to high mortality rates. Therefore, early diagnosis and prompt, aggressive medical and surgical treatment are paramount.
Surgical debridement, fasciotomies and unroofing of infected tracks are the cornerstones of the treatment, removing all infected necrotic tissue is imperative in the face of an infected DFA. The key to success is the correct appreciation of the extent of infection, based on the knowledge of the anatomical pathways for infection spreading through the tendon sheaths and through compartments, as well as adequate debridement to decrease infected tissue burden (Capobianco et al, 2010; Carro et al, 2020). Once the DFA has been detected, and the initial conditions of the patient have been evaluated, the selected antibiotic scheme is started, submitting the patient to surgical debridement within the first 24 hours, especially if C-reactive protein is >100 mg/l, and a new surgical debridement is considered in 48 hours if there is no clinical improvement (Aragón-Sánchez et al, 2012). Areas with a high index of suspicion for infection, such as the presence of fluctuations or crepitus in tissues (even without having an X-ray to corroborate gas in deep tissues) must be explored. Crepitus is an indication to extend the dissection until healthy tissue is reached.
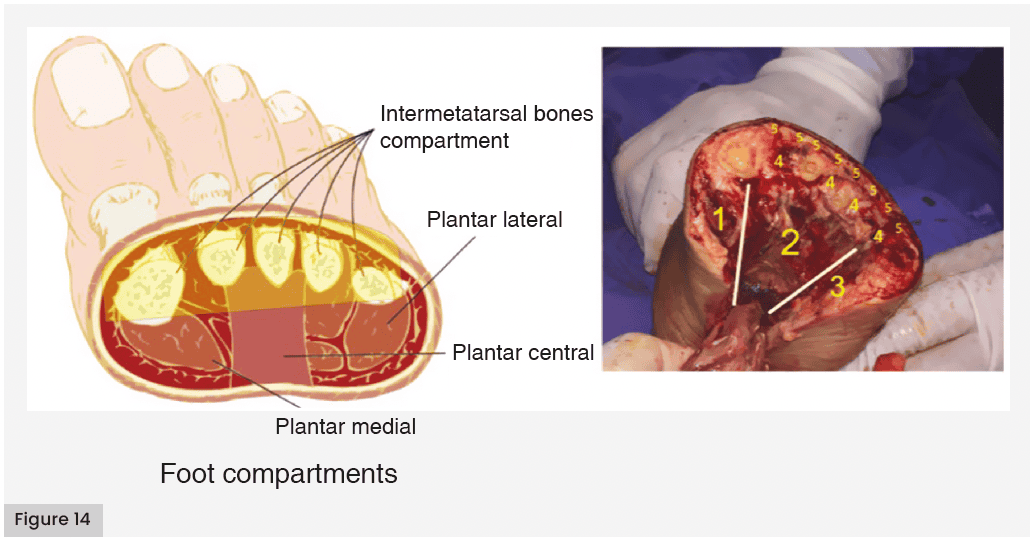
The fascias are firm tissues that divide the foot into five compartments, which are especially relevant in the spread of the infection and in the presentation of the clinical picture.
The plantar fascia forms three compartments with rigid characteristics, one central and two laterals, these are closely related to the interosseous compartment, which is the space between the metatarsals, above this is the dorsal space [Figure 14].
The presence of osteomyelitis in DFA due to infection makes it necessary to perform bone curettage during the initial debridement. Although there are studies of conservative management, this is inadequate in the presence of necrotising fasciitis, septic arthritis and disseminated infection in deep tissues. A necrotic infected bone is identified by colour changes, dark appearance, necrosis, it does not bleed and breaks apart easily; and it must be removed until bone of a firm consistency is reached and with bleeding, taking a sample of the remaining bone for culture and antibiogram.
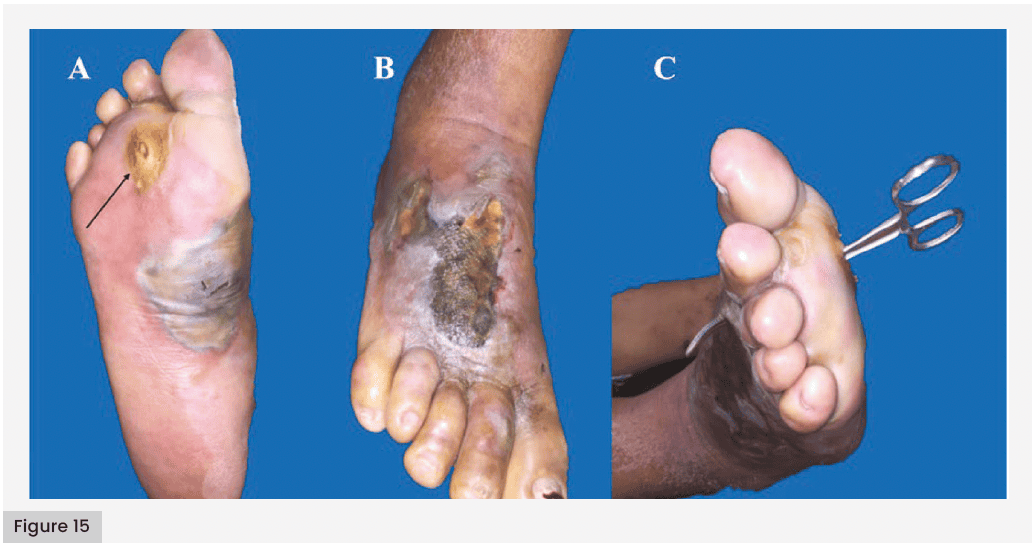
Sometimes, the entry site is easily identified, such as a neuropathic ulcer on the metatarsal head that reaches the deep compartment through tendon infection [Figures 15, 16 and 17]. On other occasions, the entry site may not be visible, as occurs in those patients who suffer a foreign body lesion (a typical example of a nail lesion in a neuropathic foot) where the bacteria reach the deep compartment inoculated by the nail and proliferate. Initially, symptoms may be absent; However, within a few days, the onset of inflammation and accumulation of purulent material significantly increases pressure within the compartment. This leads to compression of vascular structures and necrosis of distal tissues. In both cases, the infection tends to spread from areas of higher pressure to lower pressure. Due to the high resistance of the plantar fascia, the infection may extend into the interosseous space and subsequently reach the dorsal compartment. As a result, necrosis and clinical signs of infection may first appear on the dorsum of the foot, rather than at the original site of entry.
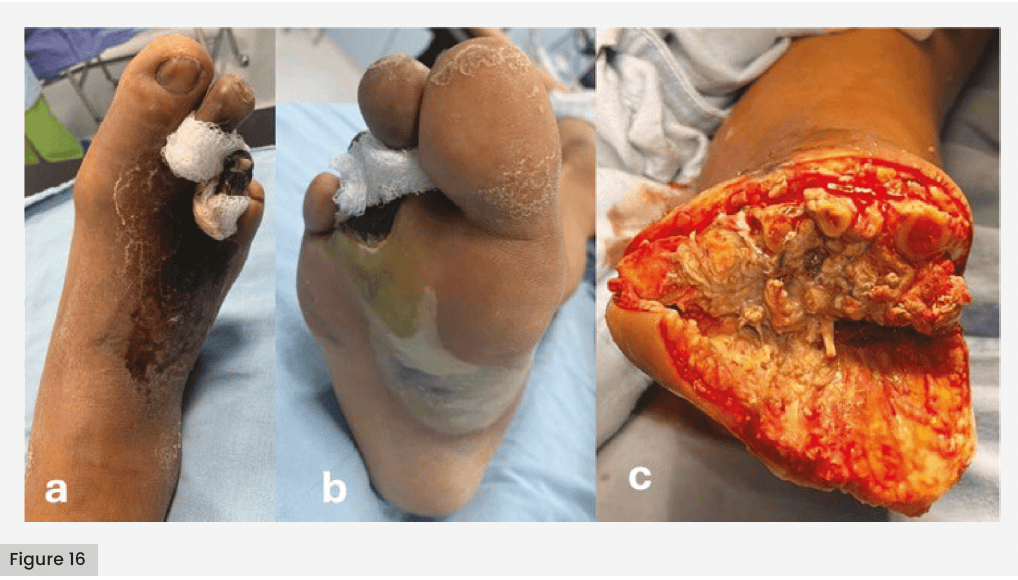
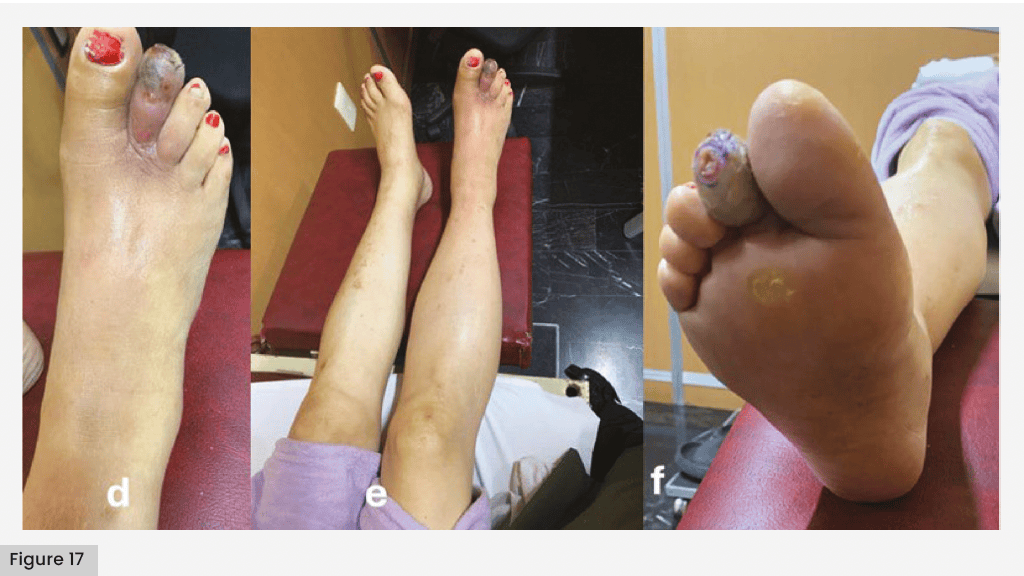
Debridement should be performed systematically, removing infected skin and fatty tissue, affected tendons, exposing tendon sheaths, and resecting affected bone; look for fistulous tracts and deep abscesses; in the presence of irreversible digital necrosis, consider amputation of the affected digits. If the irreversible necrosis involves all the digits or the forefoot, use amputations at different levels, such as transmetatarsal, Lisfranc or Chopart amputations.
When ischaemia is the cause of a single foot attack, it is preferable to wait for revascularisation before debridement of the wound; however, when the acute attack is mixed (infection and ischaemia), debridement is a priority to control infection and subsequently undergo a revascularisation protocol, preferably within 48 hours (Thiruvoipati et al, 2015).
The healing and cleaning of wound care will continue, debriding as many times as necessary. After debriding the entire infected necrotic area, the wound should be washed with saline, checking that there are no areas that went unnoticed.
3.2.1.3. Treatment of ischaemic DFA
Critical ischaemia is the most devastating presentation of DF syndrome independent of the severity grade, with or without tissue loss, with recent acute deterioration. Without prompt treatment, it can escalate to limb-threatening proportions in hours, and poses an elevated risk of amputation and mortality. The ischaemic DFA is equivalent to an acute myocardial infarction or a cerebral vascular event, where ‘time is tissue’; early recognition is required, aggressive management and revascularisation is urgent, either by conventional or endovascular methods (Vas et al, 2018; Carro et al, 2020). The Latin American SEWSS, allows the graduation of ischaemia by degrees of severity, as well as the initial evaluation and follow-up, which is relevant in its prognosis [Figure 18] (Martinez-De Jesús, 2010a; Martinez-De Jesús et al, 2022).
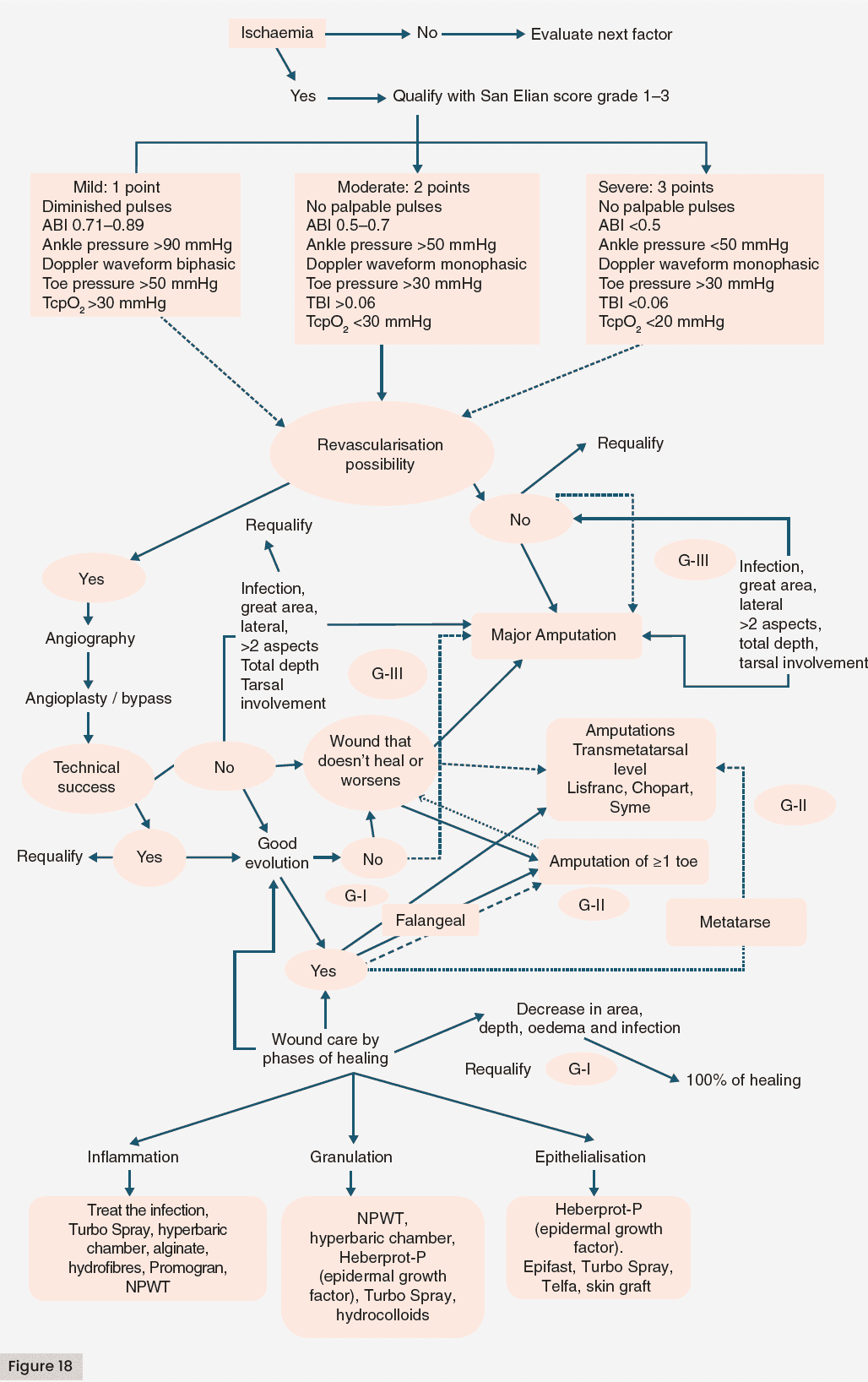
Diabetes confers an abnormal metabolic state that favours accelerated atherosclerosis in the vascular tree, including a proinflammatory state associated with cellular structures of the vessels, with a predominance of endothelium and smooth muscle. Lesions in patients with diabetes are diffuse, distal and multisegmental, mostly affecting the infrapatellar system, and in some patients, calcification of the medial arterial layer occurs, called Mönckeberg calcification (Martinez-De Jesús, 2010a; Carro et al, 2020). Clinical presentation may include pain on walking; pain at rest of the foot; shiny, sometimes erythematous skin; hair loss; distal coldness; thickened nails; Buerger’s sign; delayed capillary refill of >2 seconds; and areas of necrosis (Martinez-De Jesús, 2010a). Often, they may appear as small lesions, such as a ‘black toe’; however, one of these lesions represents a potential risk for the loss of the limb. In ischaemic DFA, the posterior pedal and tibial pulses are found barely palpable or not palpable, and the haemodynamic tests are altered, corresponding to grade 2 (moderate) or grade 3 (severe) of the ischaemia classification according to SEWSS (Martinez-De Jesús et al, 2022).
Critical limb-threatening ischaemia represents the end-stage of peripheral
arterial disease. In this stage, there is a great risk of mortality, loss of the limb, pain and a
very limited quality of life (Conte et al, 2019). Multiple health professionals are involved
in the management of the DF; if a systematisation in its approach is not carried out, its diagnosis can be delayed or ignored. The non-invasive vascular laboratory should be performed by the medical personnel of the first level of care (first contact doctor, nursing, podiatry) up to the third level of specialty (vascular surgery, general surgery, traumatology, rehabilitation, radiologists and interventional cardiologists, podiatrists, internal medicine, endocrinology). Within the vascular physical examination of the lower extremity, pulse palpation is included, which should be a routine practice in all patients with diabetes.
Patients should be examined at least once a year for those with foot risk category 0 (without arterial disease), and every 6–12 months in category 1 (arterial insufficiency), according to the International Working Group on the Diabetic Foot (Fitridge et al, 2024).
Restoration of foot perfusion is only one part of treatment, which must be provided by a multidisciplinary care team. Therefore, any revascularisation procedure should be part of a comprehensive care plan.
In patients with deep infection, such as an abscess of the foot, infection of a deep compartment of the foot that needs immediate drainage or extensive tissue loss/gangrene that must be removed to control infection, immediate drainage should first be considered to control sepsis.
This must be accompanied by aggressive antibiotic therapy, initially broad-spectrum, and rationalised according to tissue cultures – “time is tissue” in these patients (Carro et al, 2020).
A study carried out at the National Institute of Medical Sciences and Nutrition Salvador Zubirán, from the Section of Angiology and Vascular Surgery in Mexico City concluded that part of the high number of amputations, despite revascularisation procedures, was due to the lack of early referral to the vascular surgeon and the absence of a system to appropriately evaluate patients with DF (Hinojosa et al, 2018). Evaluation of the arterial tree is essential to consider the possibility of rapid revascularisation (i.e. within a few days). It must be decided who needs arterial revascularisation of the lower extremity, and which procedure is the most appropriate to achieve this must be determined. For the vascular surgeon, the use of the Global Limb Anatomic Staging System classification is recommended for this objective (Elgzyri et al, 2013).
All diabetes patients with foot injuries of ischaemic aetiology should be referred to a more complex centre, so that revascularisation procedures of the affected extremity are performed as soon as possible. Revascularisation requires appropriate imaging to guide therapy. It is unacceptable to rely only on clinical examination before performing a revascularisation procedure. Anatomical information about the arteries of the lower extremity should be obtained to assess the presence, severity and distribution of arterial stenosis or occlusions. Health professionals should be aware of these techniques and their limitations in individual patients. The decision on which imaging modality to use will depend on the patient’s contraindications, as well as local availability and experience.
It is important to recommend systematic planning of lower-extremity revascularisation, considering it successful when this procedure produces low post-procedure morbidity and mortality, when there is preservation or restoration of the patient’s independent ambulation, improves their quality of life, and has a lower cost for the health system.
In Latin America, there are few reports regarding the results of open or endovascular revascularisation techniques, depending on the country and even the geographical area investigated. A study published by the Angiology, Vascular and Endovascular Surgery Service of the National Medical Center Mexico City reported rates of up to 95% of salvage at 6 months with infrapopliteal endovascular surgery (Lecuona-Huet et al, 2013).
It is suggested that the technique to be used considers as many technical aspects of the operator, as well as the availability of supplies and equipment; however, revascularisation treatment should not be neglected for those who need it. Although most patients require a single procedure to achieve this, diabetes patients also require debridement procedures and minor amputation surgeries for resection of infected and ischaemic tissue (Conte et al, 2019).
Revascularisation should not be performed if there is no real possibility of wound healing, or when major amputation is unavoidable. Many patients are at high anaesthetic risk due to comorbidities, and bypass surgery confers a significant risk of perioperative complications. Patients who may not be suitable for revascularisation are frail, with short life expectancy, have poor functional status, confined to bed, have a large area of tissue destruction that makes the foot functionally unsalvageable and those who cannot be mobilised after revascularisation (Fitridge et al, 2024). The decision to proceed with primary amputation, or palliative care, must be made jointly with the patient and the healthcare team. Primary amputation in patients with critical ischaemia or life-threatening DF injuries is defined as lower-extremity amputation without a prior history of revascularisation procedures. Revascularisation should be avoided in patients in whom, from their perspective, the risk–benefit ratio for the probability of success of the procedure is unfavourable.
Primary amputation should be offered to ischaemic patients who have pre-existing lower-extremity dysfunction (uncorrectable flexion contracture, paralysis) or the foot is unsalvageable, poor functional status (e.g. bedridden) or short life expectancy.
For those patients in whom the risk–benefit relationship of revascularisation is not clear, it should be considered that some ischaemic wounds heal without revascularisation. One study found that in selected patients with mild-to-moderate ischaemia and tissue loss, a multidisciplinary, stratified approach can achieve acceptable rates of wound healing and limb salvage with a limited need for delayed revascularisation (Gabel et al, 2020).
There is no evidence on the use of prostaglandins or other types of agents to manage acute ischaemic attacks, so they should not be recommended.
The following are some recommendations on the treatment of ischemia in DFAs.
- DFA due to ischaemia is the most devastating presentation of DF syndrome, where “time is tissue” and revascularisation is urgent.
- The ischaemia classification of the Latin American SEWSS by degrees of severity is relevant in its prognosis.
- Non-invasive vascular assessment must be carried out at all levels of care and to the extent that resources allow it, making it necessary to refer the patient early to a higher level of care to resolve the problem in an adequate time.
- Non-invasive study methods accurately report the degree of ischaemia of the foot and the anatomy of the vascular tree. It is advisable not to use contrasting studies prior to these studies.
- Follow the Global Limb Anatomic Staging System classification for the indication of revascularisation and its type.
- The technique to be used for revascularisation should consider technical aspects of the operator, as well as the availability of supplies and equipment. Offer treatment to those who need it.
- After foot perfusion restoration, the patient should be provided with the care of a multidisciplinary team for comprehensive care.
3.2.1.4. Treatment of DFA secondary to oedema
In most cases of oedema and wounds, we must always look for the underlying cause or the comorbidity that the patient is presenting at that moment and direct the treatment towards that objective. If the patient is on renal replacement therapy, you should evaluate the effectiveness of haemodialysis or peritoneal dialysis. Patients with congestive heart failure should be compensated and treated with diuretics to reduce oedema to better treat the ulcer or wound for which the patient is consulting.
Several general measures can be employed to manage oedema in DF patients. These measures include reducing salt intake and adopting a low-sodium diet, elevating the extremities, using compression stockings to aid venous return, and considering the use of diuretics. In some cases, it may be necessary to evaluate serum albumin and total protein levels to improve albumin levels and, consequently, the oncotic colloid pressure of the plasma.
Despite the numerous existing classifications for the management of DF conditions, oedema has often been overlooked as a significant variable to consider. However, its presence can impede the healing process of ulcers, particularly when compartment syndrome is associated with the wound or ulcer. In such cases, drainage, unroofing or fasciotomies may be required for release. It is important to highlight that oedema occurs in 38% of DF patients, 58% of those who require amputation and 5% of those who die (Apelqvist et al, 1990; Ho and Leigh, 2012). Notably, reducing oedema through innovative pump technology has been shown to improve healing rates by 20% compared with placebo (Armstrong and Nguyen, 2000).
In the context of the SEWSS, oedema is assigned 1 point if it is present in the area of the ulcer or wound, 2 points if it affects the entire foot or extends up to the ankle and 3 points if it is a consequence of a general disease [Table 3].
Oedema is an initial component of the normal acute inflammatory response process that can become chronic due to failure of cellular and tissue resorption processes. The maintenance and/or presence of intracellular oedema is one of the main causes of failure in ulcer healing, the treatment of oedema in the DF is a critical intervention to prevent serious complications, such as infections, chronic ulcers and, in severe cases, amputations (Apelqvist et al, 2008).
Unilateral oedema due to sepsis or neuropathy may be treated with Jones bandages or newer technology, or may progress to infectious lymphangitis, requiring more complex therapeutic approaches. Secondary oedema is considered the most serious, as it results from a chronic underlying condition that is difficult to control. In cases where the oedema is refractory to conservative treatment, or if there is severe ischaemia that puts the viability of the foot at risk, it may be necessary to consider surgical interventions (Singh et al, 2005).
The treatment of DFA caused by oedema involves first diagnosing the underlying cause of the oedema quickly and accurately [Figure 19]. The treatment should be tailored based on the specific pathology affecting the patient. It is essential to conduct a differential diagnosis to identify if the oedema is cardiac-related, such as heart failure or pulmonary oedema; hepatic-related, such as cirrhosis and secondary ascites; or renal-related, particularly chronic kidney disease, which is the most common cause of lower-limb oedema in patients with long-standing diabetes. Additionally, venous insufficiency or lymphoedema should be ruled out. Preventing kidney damage is crucial to avoid complications.
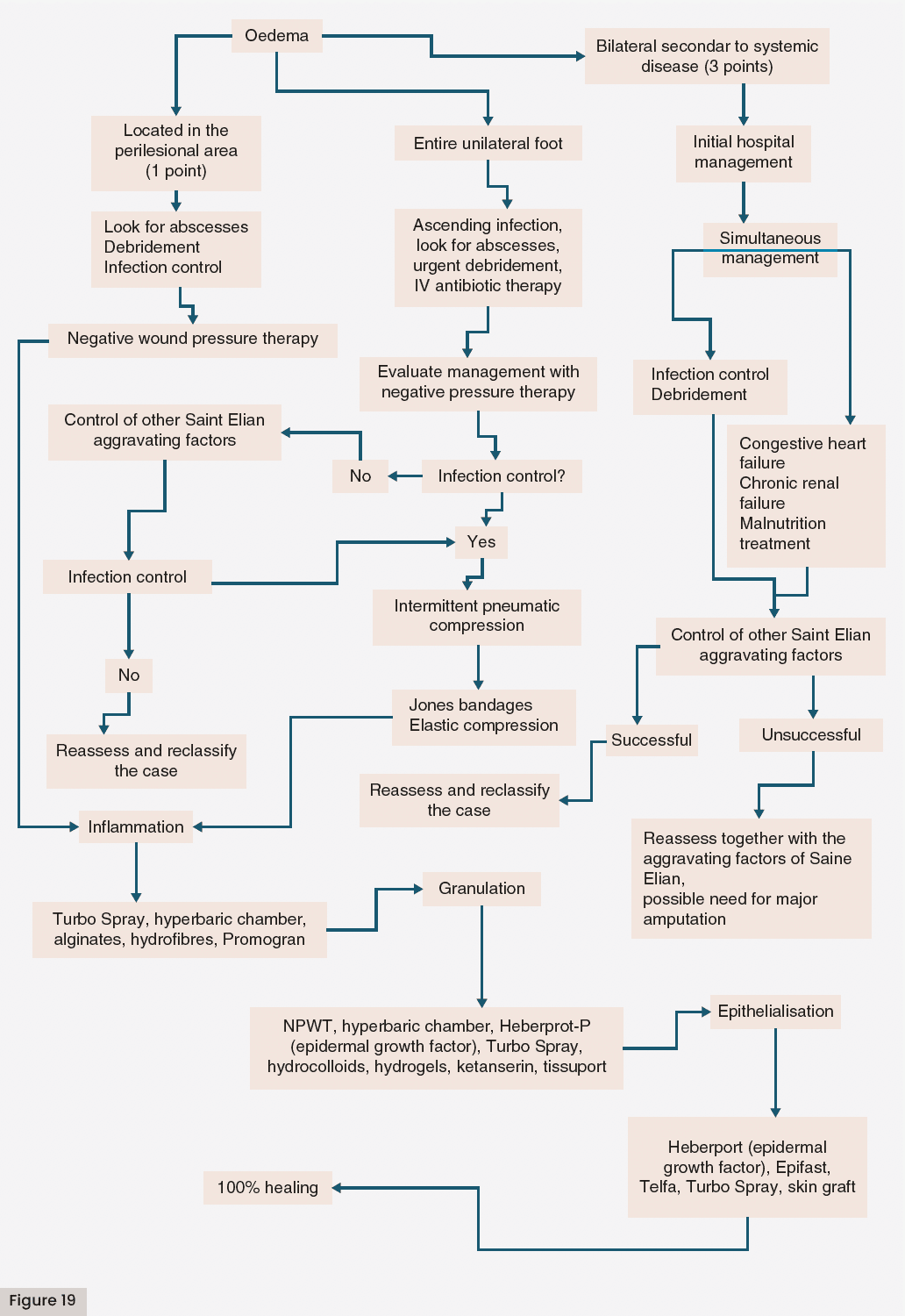
A low-fat and low-salt diet is recommended, along with maintaining blood glucose levels within a normal range, individualised daily exercise, weight control and monitoring blood pressure. Besides the necessary medical care to address the diseases causing oedema, the following measures can help alleviate swelling in the legs, ankles and feet:
- Elevating the legs above heart level while lying down.
- Avoiding prolonged sitting or standing without movement.
- Engaging in walking or exercise that facilitates fluid drainage from the legs back to the heart.
- Wearing supportive elastic stockings that provide pressure, and prevent fluid accumulation in the legs, ankles and feet.
- Adopting a low-salt diet.
- Taking diuretics (as appropriate) to help eliminate excess salt and fluids from the body.
- Assessing low levels of albumin and total proteins.
- Protecting the swollen area from extreme temperatures, injuries or pressure, as any injury will take longer to heal and may become susceptible to infection.
For the treatment of DFA due to oedema, in addition to the measures mentioned above to control excess fluid in the legs, it is essential to take care of the wound area. Most of these wounds have a moderate-to-elevated level of exudate, promoting the growth of bacteria, such as Pseudomonas aeruginosa and methicillin-resistant S. aureus, as well as fungi, such as Candida albicans. The oedema should be reduced first, followed by reducing wound moisture using alginate silver dressings, which have high absorption capabilities, reduce moisture on the skin and aid in better healing of wounds in the epithelialisation phase.
Negative pressure wound therapy (NPWT) simplifies or shortens the wound healing periods due to its extensive mechanism of action, favouring biological processes involved in the healing process. Among the mechanisms documented in the literature are:
- Increased blood flow or neovascularisation: Mechanical forces exerted by NPWT control stimulate cell mitosis, leading to angiogenesis. Neovascularisation allows for the recruitment of neighbouring tissues to the wound area, stimulating adjacent tissues.
- Oedema reduction: NPWT pressure allows for fluid removal, and decreases peripheral oedema, promoting progressive microvascular circulation, and improving the wound bed by increasing the elimination of exudates containing proteases and their degradation products, as well as growth-inhibiting factors.
Managing this type of DFA remains challenging, as addressing oedema is a fundamental part of wound healing and resolution. Accurate diagnosis and comprehensive management can yield favourable results for patients [Figure 14].
3.2.1.5. The DFA secondary to neuropathy (Charcot foot)
Charcot neuroarthropathy (CN), although uncommon, it is a potentially devastating late complication of diabetic neuropathy [Figure 20]. CN occurs in a well-perfused foot with both somatic and autonomic neuropathy; the patient presenting with acute CN tends to be slightly younger than is usual for those presenting with foot ulcers. There may be a history of trauma, although it may be overlooked because of severe sensory loss.
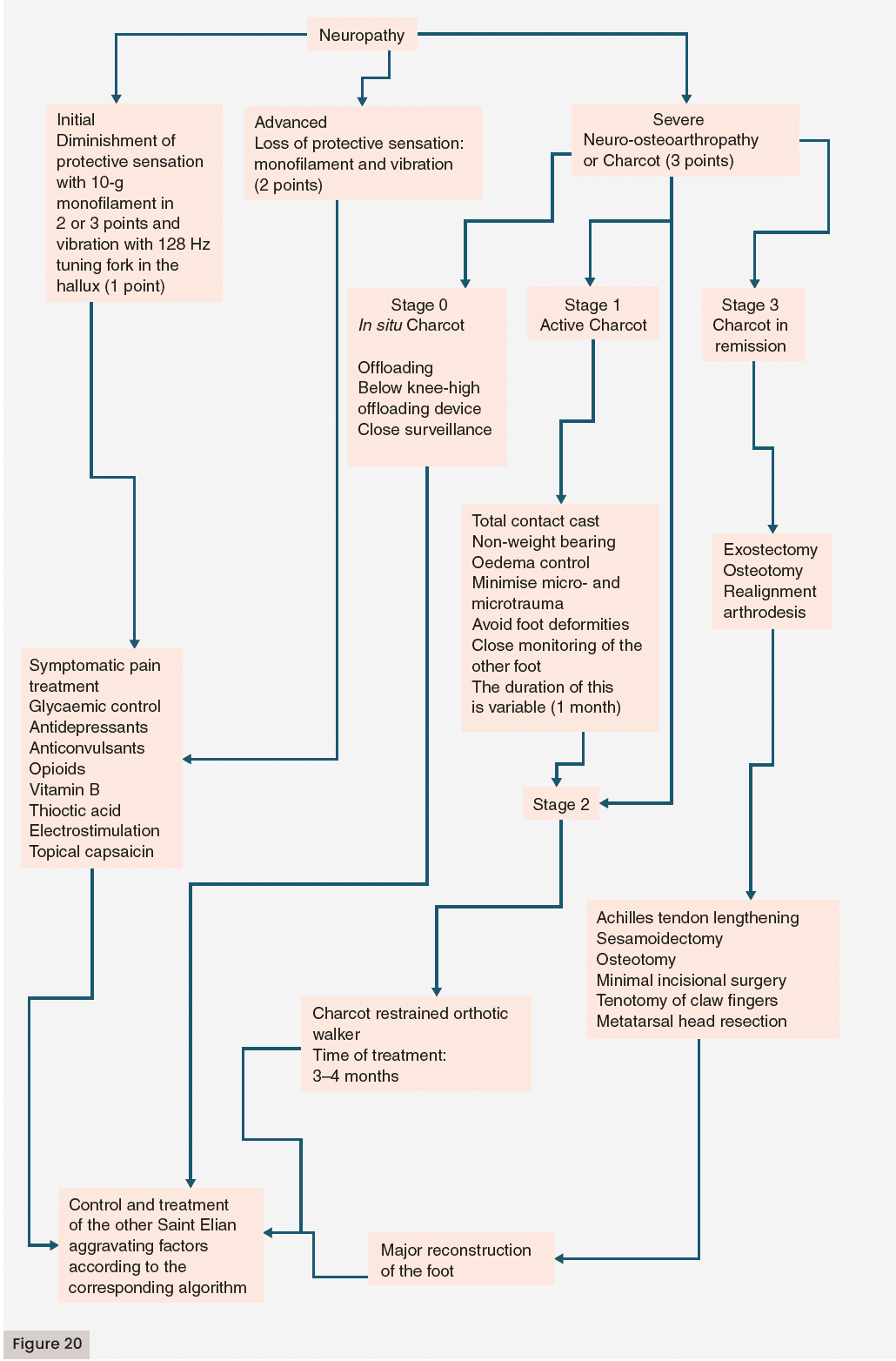
The most important issue in the treatment of this condition is the recognition of acute Charcot foot. Any patient with known neuropathy who presents with a hot and swollen foot of unknown cause should be assumed to have acute CN until proven otherwise.
Diabetes is a chronic disease that produces neuropathic, sensory, motor and autonomic alterations, which, associated with macrovascular and microvascular disease, and biomechanical alterations, will produce an acute mixed attack over time in a diseased DF.
Charcot foot treatment goals:
- To reduce the foot and ankle destructive effects.
- Delay the late stage of foot deformity.
- Offloading is the most important initial treatment.
- Surgery: Bone exostectomy with/without wound arthrodesis, and internal and external fixation. Bone resection when osteomyelitis is part of an acute attack. Do not perform reconstructive surgery during the acute attack.
3.2.1.5. Mixed DFA
For many years, researchers on DF have been trying to draw attention to the need for timely management of DF crises. Unfortunately, this lack of timely intervention and the burden on healthcare services often result in these patients requiring a major amputation.
The DFA, or acute DFA, is a definition published by Vas et al. (2018) to emphasise the importance of the ‘golden hour’ in the urgent management of this issue, similar to the presentation conditions of an acute myocardial infarction or a cerebrovascular event. As mentioned in their writing, ‘time is tissue’, and the more time that passes during the course of the acute attack, the greater the tissue loss the patient will experience. Their overall clinical evolution will also be more complicated, leading to the loss of the limb and even putting their life at risk. We can define DFA into five categories: infectious, ischaemic, neuropathic, oedematous and mixed, the latter being the topic that we will develop, since each category was specifically developed in previous chapters (Martinez-De Jesús et al, 2021).
The mixed DFA is characterised by the presence of two or more aggravating factors, which are systematised in the SEWSS: infection, ischaemia, oedema or neuropathy [Table 3] (Martinez-De Jesús, 2010a; Martinez-De Jesús et al, 2021).
In the mixed attack, any of the three degrees of condition that present the four aggravating factors can occur; for example, it can be a patient with grade 3 ischaemia (ankle-brachial index <0.5, toe-brachial index <0.6, finger pressure <30 mmHg), together with grade 2 or moderate infection (cellulitis >2 cm, abscesses, necrosis, fasciitis, osteomyelitis and/or arthritis), with grade 2 oedema (unilateral of the foot or extremity) and with grade 2 neuropathy (advanced, lack of protective sensitivity to pressure and vibratory) [Figure 5].
It means that in mixed DFA, 256 different clinical findings can be found based only on aggravating factors, being the most complex DFA and for which very little has yet been referred to or written about in the scientific literature.
3.2.1.6.1. Renal attack (renal foot) as a prototype of mixed DFA
In the understanding that two or more aggravating factors can be found in mixed DFA, we consider that renal attack (or renal foot) is the prototype of this clinical spectrum, since this type of patient with chronic kidney disease (CKD) can present with some degree of ischaemia, infection, neuropathy and oedema (mainly due to renal failure).
Within the pathophysiology and medical history, we could consider the following features in the renal foot:
- Patients with diabetes and CKD have a threefold greater risk of presenting a foot ulcer or wound and a sevenfold greater risk of having an amputation (Ndip et al, 2010).
- Patients with CKD have worse metabolic control and a persistent inflammatory state secondary to hypoalbuminemia/malnutrition, systemic inflammatory reaction (elevated protein C) and elevated HbA1c. These metabolic findings have been associated with a delay in the risk of infection and in wound healing.
- Patients with peripheral neuropathy and CKD have a threefold greater risk of developing an ulcer or wound and presenting a subsequent amputation (Kaminski et al, 2015).
- Patients with end-stage renal disease are at greater risk of loss of protective and vibratory sensitivity due to uremic polyneuropathy. They also have a greater risk of developing autonomic neuropathy, causing greater dryness of the skin, the development of fissures and the subsequent appearance of wounds and ulcers.
- Patients with CKD develop higher plasmatic phosphorus levels (>0.40 mg/dl), secondary to a higher degree of vascular calcification medial layer arterial calcification and the appearance of small-vessel arterial or microcirculation. These findings can aggravate the development of peripheral arterial disease and confer a greater risk of developing an amputation (nine-fold greater risk of presenting an amputation; Kaminski et al, 2015). It has been seen that patients on haemodialysis present defective microcirculation secondary to dialysis management during and 4 hours after the haemodialysis session (this can aggravate or cause greater peripheral arterial involvement).
- Patients with renal foot have a higher degree of stenosis–occlusion of the posterior tibial artery and the lateral plantar artery, making it more difficult to manage by open revascularisation or angioplasty (Baghdasaryan et al, 2020). Patients with renal foot have a higher risk of developing small-vessel arterial or microcirculation medial layer arterial calcification, causing greater obstructive arterial disease at the level of infra-popliteal vessels and at the level of vessels of the foot. This can lead the patient to critical ischaemia, without having options for revascularisation by open or endovascular surgery (no-option critical ischaemia).
Mixed DFA is a complex presentation due to all the variants that cause the severity factors (ischaemia, oedema, infection, neuropathy), and can occur in the patient. It is imperative for the clinician to have a solid knowledge of the SEWSS, and have the basic tools to assess each of these factors (linear Doppler, tuning fork). In the same way, it is necessary to carry out laboratory studies (haematic biometry, blood chemistry, serum electrolytes –including phosphorus and plasmatic calcium) to assess the metabolic state of the patient and its infectious severity (leucocytosis, leukopenia, elevation of C-reactive protein) in case it presents this aggravating factor. Vascular assessment is important, and emphasis should be placed on the clinical data that make up the ischaemia severity index, to be able to stage the patient in this aggravating factor and refer them in a timely manner to the vascular surgeon, who will complement the clinical and laboratory studies, and will plan the open or endovascular revascularisation procedure with a study of angiotomography or angioresonance.
Based on the diversity and complexity of mixed AAPD, management is multidisciplinary, and, like any acute attack, it must be prompt and timely. A flowchart for treatment is suggested in the following scheme.
3.2.2. Acceleration of wound healing phases
The SEWSS is used for diagnosis, prognosis, treatment, prevention and follow-up, with a system of evaluation and treatment, and chronobiology that can determine that a very serious case has an improvement in results, successful healing time, and reduces the rate amputations and mortality according to the following protocol:
- Triage: Identifying the priority and severity of the case.
- Management of aggravating factors: Implementing measures that promote limb salvage and wound healing.
- Acceleration of healing: Using various tools and devices for wound care.
- Homeostasis: Metabolic, nutritional and exercise control.
- Rehabilitation: Follow-up and care for the limbs to reduce the possibility of new acute events and manage chronic conditions without complications.
For advanced wound care, we currently use various tools to keep wounds clean and in favourable condition for healing under the systematic re-evaluation using the Latin American SEWSS follow-up sheet that allows us to objectively assess wound progression or regression. The MOIST system provides a reasonable order of these different interventions to accelerate wound healing (Dissemond et al, 2024).
According to the purpose of these recommendations, we comment on the available documented strategies in Latin America as we do it.
The acronym MOIST includes: moisture balance, oxygen balance, infection control, supporting strategies and tissue management.
3.2.2.1. Moisture balance
Examples of types of wound dressings depending on the amount of exudate are as follow: low exudate; hydrogels, hydrocolloids and semipermeable films; moderate exudate: foams, alginates and fibres; and high exudates: foams, fibres and super absorbers.
3.2.2.2. Oxygen balance
The therapeutic approaches with oxygen for local therapy are listed in Table 8 according to Gottrup et al. Efficacy of hyperbaric oxygen therapy is controversial, but is currently used in many countries in Latin America. Local wound delivery of oxygen is applied in some centres as boot modalities or by direct wound delivery devices.
3.2.2.3. Infection control
The measures for infection control can only be started after the resolution of the foot attack, the necrotising fasciitis and the compartment syndrome with immediate surgery. Then, a scheduled protocol to clean wounds with physiological saline, surgical soap and additional tools must be initiated. We reassess wound healing conditions at each dressing change, looking for reduced wound area, wound contraction, decreased oedema and good granulation.
Physiological saline and oxygen with Turbo Spray, hydrojet or Jetox are used. This allows for gentle debridement and maintains the wound in good condition, speeding up the healing process. Super-oxidised solutions can be helpful, but are not indispensable. Wound dressings are used judiciously to keep the wound moist, preventing desiccation. There are various brands on the market; these vary from country to country.
3.2.2.4. Supporting strategies
Examples of actions that are already used in Latin America to actively support wound healing are: cytokines, growth factors, macrophages, matrix metalloproteinases, pH values, reactive oxygen species and stem cells.
3.2.2.4.1. The recombinant human epidermal growth factor (Heberprot-p)
Diabetic foot ulcers (DFUs) require a long time to heal and can become chronic, associated with poor glycaemic control, refractory infection, anti-inflammatory cytokines and vascular injury, primarily due to decreased levels of epidermal growth factor (EGF) and its specific receptor. It has been shown that the intralesional and perilesional application of EGF in the wound, as the only injectable medication adjuvant to the specific treatment of DFU, can promote wound healing, gliocyte and fibroblast proliferation, neo-epidermal thickening, peripheral nerve regeneration, and mediate the proliferation, migration, and differentiation of gliocytes and fibroblasts, improving granulation and stimulating epithelialisation (Fernández-Montequín et al, 2009a; Zhang et al, 2019). A decrease in the levels of proinflammatory cytokines has been observed, suggesting that EGF not only promotes tissue regeneration, but also modulates the inflammatory response, favouring a more conducive environment for healing (Ojalvo et al, 2016). Although one publication reports the reduction of the risk of unnecessary amputation by up to 70%, we use and recommend it as an adjuvant therapy to enhance granulation tissue in 86% of patients with DFUs, and achieve complete ulcer closure in up to 75% of patients (Fernández-Montequin et al, 2009b; López-Saura et al, 2013; Martínez-de Jesús, 2024).
Intralesional infiltration of EGF has been associated with a significant recovery of markers of oxidative stress and antioxidant reserve, contributing to restoring the systemic redox balance, with intralesional and perilesional application (Cacua Sanchez et al, 2023). The appropriate EGF for application should be recombinant human EGF, which is included in the Colombian Guide for the Management of Complicated Diabetic Foot Ulcers (developed by 9 scientific associations) and in the ‘Recommendations for the Management of Patients with Diabetic Foot – Instructional Course’, on behalf of the Colombian Society of Orthopaedics and Traumatology (Asociación Colombiana de Diabetes, 2019; Triana-Ricci et al, 2021). It has also been seen that EGF is cost-effective, with respect to both conventional therapies and more eso-specific therapies, such as negative pressure therapy (Osorio and Jubiz, 2021).

3.2.2.4.2. Cryopreserved allografts of human epidermal keratinocyte cultures
Cryopreserved allografts of human epidermal keratinocyte cultures (Epifast) promote cellular migration and produce growth factors that stimulate cell proliferation from ulcerated skin to achieve re-epithelialisation.
The application of tissue-engineering technology to wound healing has become an option for the treatment of DFU. Martínez-De Jesus et al (2022) published a comparative, prospective study conducted to assess the efficacy of Epifast to enhance wound healing in granulating DFU. Epifast displayed a shorter duration of the epithelialisation phase and upon the entire wound healing process than standard treatment, reaching wound closure 50% faster. Patients under standard treatment without Epifast displayed a slower healing as the SEWSS severity grade increased. Diabetic foot wounds treated with Epifast displayed a shorter time to complete re-epithelialisation than wounds treated with standard care.
Other biological products, such as amniotic membrane, adipose-derived or mesenchymal stem cells, can also be used. In this context, other skin substitute products for wound treatment can be mentioned.
3.2.2.4.3. pH control and reactive oxygen species pH superoxidised solutions modalities containing reactive oxygen species
The use of antiseptics in wound care is often controversial, and there is a need for a non-toxic, highly disinfectant agent. Martínez-De Jesus et al. assessed the efficacy of a neutral pH superoxidised aqueous solution for infection control, odour reduction, and surrounding skin and tissue damage on infected DF ulcerations. Odour reduction was achieved in all patients treated with neutral pH superoxidised aqueous solution and surrounding cellulitis diminished. Meanwhile, granulating tissue increased. Neutral pH superoxidised aqueous solution, as part of a comprehensive care regimen, may be more efficacious in infection control, odour and erythema reduction than conventional disinfectants in treatment of DF infections.
3.2.2.5. Tissue management
For further wound treatment, neutral wound dressings or physical devices, such as NPWT, can subsequently be used. NPWT is an active wound management system that utilises controlled negative pressure, which is applied uniformly to the wound through an open cell foam or other interface dressing in a continuous or intermittent fashion. With NPWT, after infection is controlled, these sponges connected to a device producing controlled negative pressure can be used to maintain hypoxia of the wound and promote fibroblast proliferation, supporting wound contraction. Cost considerations may limit their use, but they are excellent if the equipment is available. There is now good evidence available for this wound treatment method in improving wound healing in several types of chronic wounds.
Other physical treatment options that can currently be used for wound treatment include; for example, electrostimulation, extracorporeal shock waves, concurrent optical and magnetic stimulation, low-intensity laser therapy or water-filtered infrared A-light.
Various drugs and emerging wound care equipment, such as artificial skin grafts, stem cells and hyperbaric chambers, are available for advanced wound care in Latin America; there are some products from bioengineering. In Mexico, cryopreserved allografts of human epidermal keratinocyte cultures (Epifast) were developed, and from Cuba, Heberprot-P, a medication containing recombinant human EGF was developed.
With the Saint Elian Scale through a directed clinical history we evaluate Ischemia, the
Table 9 shows the different options for debridement in chronic wounds.

Using the checklist, we can determine if the acute attack requires close and/or urgent intervention for limb salvage. Urgent non-invasive diagnosis will help decide if it necessitates urgent management in cases of critical ischaemia.
Oedema: Wounds located in areas with expanded interstitial spaces due to increased interstitial fluid may exhibit defects in microcirculation, leading to subepidermal oedema in patients with diabetic neuropathy and ulceration. This contributes to reduced skin thickness and predisposes to further lesions, delaying wound healing. Oedema is an extremely crucial factor affecting tissue oxygenation by increasing intercellular spaces, thereby increasing the distance for oxygen diffusion to more distant cells.
Neuropathy: Timely offloading of foot lesions, even if small, through comprehensive evaluation using the SEWSS, allows us to ensure proper healing. Preventive biomechanical assessments and the use of custom-made insoles, as well as appropriate footwear, serve as protective measures to prevent the occurrence of new ulcers.
Biomechanics and Rehabilitation in the Patient after Remission of Foot Attack (Stage 3)
Biomechanics is a branch of medicine that studies the structure, function, movement and mechanical behaviour of biological systems, analysing the forces in the living being that interact for body mobility. In patients living with diabetes, studying gait biomechanics is essential due to sensory and motor alterations resulting from diabetic neuropathy, and glycation of tendinous, ligamentous and muscular structures.
In the Saint Elian Evolutive Model [Figure 20], the natural evolution of the disease is divided into three stages: stage 1, a high-risk foot with no wound, but already presenting foot deformities, loss of sensitivity or some degree of arterial insufficiency; stage 2, the acute attack where the patient already has a wound; and stage 3, a high-risk or remission stage, the following are recommendations for rehabilitation, biomechanics, footwear and prophylactic surgery.
Stage 1
Biomechanical and footwear:
- Patients with normal sensitivity and no deformities should use soft and comfortable footwear.
- Patients with loss of protective sensitivity and deformity: identify pressure peaks through baropodometry to create customised insoles with discharge in exact sites at risk of ulceration and hyperkeratosis, along with tailored footwear; toe orthoses for claw or hammer toes, gel protectors, or silicone orthoses; and callus removal decreases pressure peaks by 30%, perform it as needed.
Rehabilitation:
- Assessment by a specialist in rehabilitation medicine.
- Walking, stationary bike exercises and anaerobic exercise with the use of custom insoles.
- Concentric and eccentric exercises (individualised program) to maintain strength.
- Management of neuropathy, musculoskeletal contractures and sensory management.
Prophylactic surgery:
- The goal is to eliminate pressure peaks at risk of ulceration.
- Consider surgical alternatives: tendon elongation, exostectomy, osteotomies, joint plasties or tendon transpositions.
Stage 2 or acute attack
Biomechanical, ambulation and footwear recommendations:
- Depending on the severity of the lesion and the SEWSS, weight-bearing on the affected foot should be avoided.
- As the score decreases and the wound enters the granulation or epithelialisation phase, an orthosis with discharge at the ulcer level can be prescribed for essential activities.
- Pressure acting on the ulcer should be reduced and distributed to other regions.
- Total contact cast or pneumatic walker boot with discharge on the wound without removal is the most effective form for accelerating wound healing.
- Rocker bottom shoes and/or heel rockers reduce pressure peaks on the wound by 40–60%, and are recommended as a second option.
Rehabilitation:
- Emphasis on pressure relief and ulcer protection, restoration of skin perfusion, treatment of infection, oedema, neuropathy, and use of walking aids.
- Metabolic and comorbidity control.
- Patient and family education.
- Occupational therapy and psychological intervention as part of treatment.
- Individualised exercise plan without causing fatigue, resistance exercise.
- Prescription and application of physical modalities.
Surgery recommendations for offloading
Offloading surgery is performed to achieve early epithelialisation by reducing plantar pressure peaks at the wound site. The techniques used will depend on the surgeon’s skills and ensuring that there is no infection at the site to be operated on, to avoid overload points on any metatarsals or phalanges and subastragalar instability. Percutaneous surgery techniques may be a good alternative to avoid major tissue damage, allowing surgical treatment on soft and bony tissues, such as Achilles tendon elongation, subastragalar instability correction, and Reverdin–Isham, Chevron, Scarff and Akin osteotomy for the first ray, Weil osteotomy, resection of the second to fourth ray metatarsal heads, plantar osteotomies of proximal and/or dorsal phalanges, partial or complete tenotomies for flexor or extensor tendon elongations as required, gel silicone injections, and exostectomy.
Stage 3 or remission stage
The goal is to prevent a new foot attack.
- Mandatory digital baropodometry every 6–12 months (depending on availability).
- Change custom insoles annually based on baropodometry analysis to make personalised modifications.
- Perform baropodometry within the footwear, considering a re-ulceration cutoff point with a peak pressure >2 kg/cm2 or 200 kPa.
- Depending on the type of debridement, level of amputation and biomechanical changes due to tendon loss or foot structures, custom insoles should be made using 3D scanning, plaster or foam moulds.
- Patients with minor amputations proximal to the foot (Chopart, Lisfranc, Syme) are suggested to use ankle–foot or Charcot restrain orthotic walker orthoses.
- Suggestions for effective advancement in mobility and gait after a foot lesion in remission: moderate to maximum mobility for 1–3 months after wound healing; use proper therapeutic shoes with custom insoles; gradually increase the number of steps per day (if the patient does not have severe deformity or vascular disease, they can slowly, but steadily, resume activities, increasing ~10% of the number of steps every 2 weeks); in case of severe deformity or Charcot arthropathy, the goal is to have functional steps, allowing the patient to cover necessary distances at home or in the community; avoid significant variations in daily step counts; and educate the patient with emphasis on visually inspecting their feet every day.
Rehabilitation for debridement or minor amputation
The final shape of the foot, level of amputation and biomechanical changes are highly diverse; therefore, the prescription of rehabilitation treatment should be individualised.
- Maintain mobility at least at a functional level.
- Perform weight-bearing exercise.
- Practice functional gait with the use of assistive devices.
- Apply physical modalities under the prescription of a rehabilitation specialist to establish specific objectives (thermotherapy, laser, electrostimulation).
Rehabilitation for patients with major amputation
Pre-surgical rehabilitation:
- Train muscular strength.
- Maintain joint mobility.
- Ensure optimal circulatory status.
- Initiate a programme 4–6 weeks prior to surgery.
Early post-surgical rehabilitation:
- Align segments and maintain proper posture to prevent contractures.
- Preserve mobility and strength in upper limbs.
- Conduct cardiopulmonary training.
- Manage the residual limb: ensure proper healing, apply appropriate dressing and address phantom limb sensations.
- Initiate sitting and one-legged standing with the use of assistive devices.
Intermediate post-surgical rehabilitation:
- Begin treatment outside of the bed.
- Adapt a provisional prosthesis.
- Train standing with the use of the prosthesis and reshape the residual limb.
- Emphasise appropriate dressing 24 hours a day.
- Implement interdisciplinary treatment.
Prosthetic rehabilitation:
- Conduct inter- and multidisciplinary treatment.
- Adapt the definitive prosthesis within a maximum of 2 months.
- Facilitate the patient’s reintegration into work and recreational activities.
Offloading surgery is considered a useful therapy to maintain the foot with pathomechanical deformities in remission. Its goal is to eliminate the forces that increase peak pressure and bony prominences susceptible to friction or rubbing. Therefore, the foot should be maintained as a plantigrade. The techniques used will depend on the surgeon’s skills and ensuring that there is no infection at the site to be operated on, to avoid overload points on any metatarsals or phalanges, or subastragalar instability. Percutaneous surgery techniques may be a good alternative to avoid major tissue damage, allowing surgical treatment on soft and bony tissues, such as Achilles tendon elongation; subastragalar instability correction; Reverdin–Isham, Chevron, Scarf or Akin osteotomy for the first ray; Weil osteotomy; resection of second to fourth ray metatarsal heads; plantar osteotomies of proximal and/or dorsal phalanges; partial or complete tenotomies for flexor or extensor tendon elongations, as required; gel silicone injections; and exostectomy.
Health Education
Decades of diabetes research have demonstrated that there is no effective treatment for diabetes and DF prevention without education and training by healthcare personnel. The educational process is not always or almost never developed systematically, lacking basic knowledge of educational sciences and health sciences, especially of diabetes and the lower limb. This is for the health education process to be effective and consistent in primary care services, and to be implemented at the secondary and tertiary levels of care according to the status of the patient, family or community. We understand effective health education as achieving behavioural changes in individuals, and both professionals and society, which implies that only those who learn to take care of themselves can achieve an excellent quality of life. Hence, the guidance on how to achieve the competencies for health education and DF prevention is our objective, aiming to achieve continuous improvement in healthcare processes through health education in various healthcare settings.
Objectives
The overall objective is to enable the healthcare team to acquire the competencies to design, implement and evaluate educational interventions for the self-care of patients with diabetes, who are either at risk of DF or have DF.
- Specific objectives:
- Prevent complications of the foot in patients with diabetes.
- Promote attitudes and develop habits that lead to healthy lifestyles.
- Assist in understanding external determinants that negatively influence population health.
- Reduce hospital admissions.
- Educate for better utilisation of medical assistance services in DF care.
Methodology for planning health education interventions
Context analysis
Consider the psychosocial, cultural, educational and economic factors influencing the community, and determine which of these factors have influenced the overall health of the population, families and individuals. Identifying the biological factors of individuals, families and communities helps explain actual needs, aiding in the selection of effective methods, means and procedures for educational intervention planning (Asociación Colombiana de Diabetes, 2019).
Teaching principles
Health education must be active, dynamic and participatory, employing didactic techniques, such as workshops, demonstrations, sociodramas and more. Learners develop the ability to think and act for themselves, verifying whether they have achieved self-determination for their individual or family healthcare.
Formulation of objectives
Elements of educational or learning objectives foster effective communication between learners and educators, guiding the selection of content and means for teaching. Verb (desired behaviour to achieve). What (desired content to communicate). How? How will we do it? (methods and didactic techniques). Who? The learners, age, number of people. When? When will we achieve the expected behaviour (at the end or in several sessions).
Content planning
Content planning requires careful selection of topics, chosen based on the educational and health needs of the learners.
Selection of didactic resources
Didactic resources will be selected based on the age, resources, and characteristics of both the learners and the educator. Examples include the chalkboard, flipchart, posters, slides, bulletin board, computer and anatomical models, to name a few, which the educator can use to facilitate the learners’ best understanding of the intervention content.
Preparation of the educational intervention
The educator must have a mastery of evidence-based and pedagogical content.
Learning strategies
Plan participatory activities for the learners to achieve the educational objective according to their profile.
Time scheduling
The educational intervention must adhere to a predetermined time frame in planning. Respecting both the learners’ and the educator’s time contributes to discipline and order, attributes that benefit the learning process.
Educational intervention guide according to the classification and grades of DFA by SEWSS
Preventing complications arising from DF pathology is a top priority in education, which is why the approach should be comprehensive and holistic, considering the individual as a biopsychosocial being. Emphasising the importance of individual motivation to improve the health status of individuals and their feet, every foot in a person with diabetes is at risk. The best treatment we know of to date is education, understood as the intention to change the behaviour of patients to prevent complications from DF and other equally severe complications for the individual.
SEWSS grade 1 (mild)
Nonsignificant risk for adverse outcomes.
- Educational objective: Design educational interventions for individuals starting with the disease to increase their knowledge of the disease, and treatments in a simple and concise manner.
- Educational actions: Develop skills for daily foot care. Maintain metabolic control by explaining normal laboratory values in a simple manner.
SEWSS grade 2 (moderate)
Threat or risk of partial foot loss. Prognosis reserved for correct therapy and a good biological response from the patient.
- Educational objective: Design an educational intervention according to the moderate prognosis situation to contribute to a favourable response and achieve an improvement in the score as quickly as possible.
- Educational actions: Identify the needs of the patient and their caregiver to develop the educational intervention. Explain the importance of timely medical attention. Instruct on post-operative care related to foot care. Instruct on metabolic control related to prescribed medications and diet.
SEWSS grade 3 (severe)
Threat or risk of limb and/or life loss. Prognosis independent of correct technique due to poor biological response from the patient.
- Educational objective: Through educational intervention, support the primary caregiver in managing the limb salvage process in the hospital, at home or in the clinic, as appropriate. Identify the patient’s and/or primary caregiver’s family support to analyse the context and design the educational intervention according to their needs or deficits in managing the patient. Explain the benefits of continuing medical and surgical treatment. Explain that the complication is not solely a consequence of diabetes, but a result of poorly managed metabolic control.
- Educational actions: Assist the caregiver or family in identifying the severity of the health problem, seeking care alternatives and skills to support medical treatment, and achieving limb and/or life salvage. Explain basic care, such as postoperative care, metabolic control related to prescribed medications and diet, and the effects of pharmacological treatment according to educational level and physical condition. Involve family members in the educational intervention. In cases of amputation, establish a specific educational intervention plan aimed at physical and psychological rehabilitation.
Education for patients with partial or total limb amputation
- Educational objective: Provide basic knowledge for rehabilitation through educational interventions. Explain the characteristics of human gait to understand the need for appropriate prosthetics for early rehabilitation. Explain the concepts of quality of life and limb loss, psychological rehabilitation methods and techniques, and the importance of maintaining functional patterns that may be altered, such as health perception, nutrition, metabolism, activity and exercise, and cognitive and perceptual dysfunctions. Provide preventive recommendations, such as for hyperglycaemia, smoking, hypertension and dyslipidaemia. Explain basic foot care to prevent future amputations, and provide guidance on exercising lower limbs and the importance of not walking barefoot.
General recommendations
The educational process in health education cannot be one-directional; rather, it should provoke the opening of cultural behaviours for the construction of healthy lifestyles among participants. The purpose of health education and health educators is to improve the health conditions of the population through promotion, prevention and treatment, requiring interdisciplinary educational interventions. Health education should not be approached as a spontaneous task; desired learning outcomes should be planned. The nature of the educational task in health is a helping relationship, not an imposition, where the fundamental element of the educational relationship is the learner.
Telemedicine in Diabetic Foot
Telemedicine is defined as the provision of medical care when distance is a concern, by healthcare professionals who use technology for disease management and health improvement. The applications of telehealth and telemedicine can hold value in self-monitoring the foot health of individuals with diabetes, primarily for diagnostic, therapeutic and educational purposes, aiming to enhance the efficiency and effectiveness of care, patient wellbeing, and autonomy in a world with rapidly changing socio-economic perspectives in healthcare. Several applications have been developed for this purpose, including dermal thermography, foot imaging tools and mobile phones/video or online technology. However, only a few applications in DF care have been implemented, which may be related to their psychometric properties, feasibility of use, or lack of demonstrated effectiveness or cost-effectiveness (World Health Organization, 2009).
Among all the late complications of diabetes, those involving the foot have traditionally required more in-person patient visits to clinics for wound management, debridement, offloading and many other treatment modalities. The onset of the COVID-19 pandemic has not only led to the closure of most outpatient clinics for in-person consultations, but also the inability to perform the majority of laboratory and imaging investigations. This has resulted in a paradigm shift in delivering care to individuals with DFUs.
Advantages of telemedicine:
- It can be accessed from home through a smartphone, tablet or computer.
- It is cost-effective.
- It can assist in various medical conditions, such as successfully identifying the problem and its urgency, helping to prompt the patient for an immediate hospital visit. Acute Charcot neuroarthropathy might be challenging to diagnose through telemedicine and would require proper evaluation in a hospital setting. The costs of telemedicine in DF disease treatment might be like standard care. Telemedicine’s effectiveness has been demonstrated in superficial ulcers, but data on its diagnostic efficacy in deeper ulcers are yet to be established. However, the use of DF images as an independent diagnostic tool has not proven effective (Hazenberg et al, 2020).
Limitations of telemedicine:
- Telemedicine might have limited value in the overall clinical and haemodynamic assessment of a patient with DF.
- Telemedicine images or videos might not provide an accurate estimation of DF ulcer depth.
- Dark skin colour might hinder the detection of redness and cellulitis in photography.
- Telemedicine would not aid in determining the consistency of any swelling, as this requires palpation.
- Individuals with DF disease might have associated problems, such as leucocytosis, hyperglycaemia, renal insufficiency and impending sepsis, which would be difficult to detect through an online consultation.
- For older individuals with vision, hearing and cognitive issues, a telemedicine-based assessment for DF lesions could be problematic.
- In developing countries, such as India, there might be issues with internet connectivity, hardware availability and consultation archiving, which could also have potential medicolegal implications (Osorio and Jubiz, 2021).
In future, artificial intelligence and machine learning could be used to study clinical phenotypes and foot images for improved management. However, a telemedicine consultation with appropriate images/videos, combined with an assessment of other parameters, could aid in a classification system. Low-risk patients can be satisfactorily treated at home, and high-risk patients can receive a hospital/clinic visit, followed by further follow-up.
Telemedicine enables healthcare professionals to approach their patients with more knowledge, improved wound assessment skills and greater confidence. Four key success factors identified in telemedicine use were as follows: technology and training should be user-friendly, the presence of someone at the workplace who can facilitate the intervention, the need for support from committed and responsible leaders, and effective organisational-level communication.
At the patient’s home, it is also important for the community nurse to have good access to the ulcer records and the appropriate equipment, along with sufficient consultation time for the evaluation and treatment of the ulcer. In Argentina, telehealth is considered a medical act. A high-quality virtual consultation requires a committed physician to examine the patient. Subsequently, the questions will be sent to a specialist physician as a second-opinion consultation (in two separate steps), and the specialist should respond to the consultation within 7 hours (referred to as asynchronous consultation) in its basic form. This often involves using a virtual platform, ensuring confidentiality standards, with an integrated medical history format that allows attaching complementary studies and preserving information over time. Real-time synchronous consultations can include interdisciplinary meetings (a meeting between the patient’s primary physician and specialists) or virtual meetings between the primary physician, the patient and specialists.
During synchronous or asynchronous DF care, it is crucial not to overlook factors related to poor prognosis and severity that need evaluation by the professional from the beginning. Lesion assessment is the initial step in treatment, and requires a standardised system as an essential and relevant tool for decision-making. This system should improve communication among different healthcare professionals, identify those who require specialised attention and identify those who can receive care in primary or home settings.
Incorporating a suitable clinical scoring system for DFUs into daily practice is essential, and should also be considered for use in telemedicine. This anticipates the possibilities of healing and the risk of amputation, ensuring crucial factors determining wound severity and prognosis are not omitted. It should provide a reference and counter-reference model across levels of care with a multi- and interdisciplinary approach. Evaluating the wound, predicting DFU outcomes in the early stage, dynamically monitoring wound changes and rationally directing the therapeutic program are especially important, while considering the population and facilities.
The SEWSS has been internally and externally validated through scientific methods, confirmed in systematic reviews and meta-analyses, and considered one of the wound scoring methods with external validity (Martínez-De Jesús, 2010b; Huang et al, 2015; Monteiro-Soares et al, 2015; Carro et al, 2023). This classification becomes a better predictive tool for DFU outcomes in the Latin American population. It is recommended in guidelines of Latin American countries, such as Chile8 and Mexico,9 and there are experiences with its use in Latin American countries recommended in publications, and by the International Diabetes Federation and Asociación Latinoamericana de Diabetes (Rincón et al, 2012; Brugués, 2013; Consejo de Salubridad General, 2013; Márquez and Pinilla, 2013; Federación Diabetológica Colombiana, 2014; Palma and Corral, 2015; Sistema de San Elián en las Guías de Orientación técnica, 2018; Kavitha et al, 2020; Guía Clínica para la Prevención, Diagnóstico y Tratamiento Multidisciplinario del Pie Diabético colegio médico del Perú).
Medical tools
The following four modalities are considered as medical tools that can potentially serve as telemedicine applications.
Dermal thermography
In case of a temperature difference >4°F (2.2°C) between corresponding locations on the right and left foot that persists for two consecutive days, participants were advised to contact the nurse and reduce activity until temperatures normalise.
Armstrong et al (2007) suggest that counterintuitive findings regarding higher foot temperatures in ischaemic feet are a result of altered thermoregulation influenced by both neuropathy and peripheral arterial disease.
Frykberg et al (2017) proposed a smart mat based on the telehealth concept, aiming to address limitations of other thermographic tools. The system compared the temperature profile of both feet, and using a threshold of >2.22°C difference between sites on both feet, it correctly predicted 97% of foot ulcers with an average lead time of 37 days.
Hyperspectral Imaging
Hyperspectral images are used to assess tissue oxygenation in or near the ulcer based on measured oxyhaemoglobin and deoxyhaemoglobin levels. These levels are used to calculate a healing index to determine healing potential.
Several case–control studies observed a significant reduction in oxyhaemoglobin levels before ulcer closure and in healing ulcers compared with unchanged oxyhaemoglobin levels in non-healing ulcers.
A case series analysed 21 sites that ulcerated during follow-up, demonstrating that the onset of these ulcers could be predicted using hyperspectral imaging with 95% sensitivity and 80% specificity an average of 58 days before skin breakdown became evident (Yudovsky et al, 2011).
The same research group reported in a case report that an increase in epidermal thickness (callus) was associated with a decrease in oxyhaemoglobin concentration before ulceration (Yudovsky et al, 2011).
Liu et al (2013) showed that hyperspectral images of calluses, ulcers and healthy skin patches could be automatically discriminated with 97% sensitivity and 96% specificity.
Photographic images
Van Netten et al (2017) found that evaluating DFU using a mobile phone compared with in-person evaluation (as reference) strongly supported wound debridement decisions, but had low interobserver reliability, and moderate interobserver reliability for assessing the presence of ischaemia, infection, granulation, detachment, tracking or tunnelling, wet or exudative wound, cellulitis, or erythema.
Bus et al. demonstrated that using photographic images of the foot with a home-based device enabled photographic assessment (>74%) and repeated photographic evaluations (>82%) for the presence of heavy callus, ulceration and absence of signs. This was expanded by other studies, showing good feasibility of using foot photographic devices in the home environment; patient adherence was high, referrals were based on photographic assessment, which was justified, and perceived usability was good.
Furthermore, it has been demonstrated that diagnosing foot infections is valid and reliable using photographic images combined with infrared thermography, outperforming each modality used alone.
Audio/video/online communication
Regarding the feasibility of using a mobile phone to connect home nurses and physicians to support ulcer treatment, it was reported that patients were satisfied with the treatment support, as it saved time, nurses were capable of managing technical skills and felt supported, and physicians found the equipment easy to use and feasible for remote treatment, and felt they had a solid basis for decision-making with the tool (Clemensen and Larsen, 2006).
Wilbright et al (2004) reported no significant differences in ulcer healing between weekly telemedicine video interactions and in-person treatment: in 12 weeks, 75% of ulcers healed in the telemedicine group versus 81% (P=0.546) in the face-to-face treatment group. However, concerns arose regarding the lack of multidisciplinary wound care teams, patient responsibility and patient–doctor interaction.
The value of focus groups and individual semi-structured interviews of patients and healthcare professionals was investigated. Patients from both the telemedicine and control groups mentioned that superior wound care depended on a combination of competence and professional skills in wound management and continuity of care.
Clinical implications and future perspectives
If feasibility, effectiveness and cost savings are demonstrated, successful implementation of telemedicine approaches can enhance patient mobility, autonomy and health-related quality of life, especially for those who live alone, in rural areas, have cognitive issues, visual or physical impairments, or lack knowledge about the disease. This empowers patients and encourages them to take responsibility for managing their DF disease.
A small and user-friendly handheld device that can measure local foot temperature, take foot photographs and automatically process data through intelligent algorithms, which feeds back data to the patient when action is needed, is likely not far from development.
These tools, when proven viable and cost-effective, can have a significant impact on DF patient care. The application of telehealth and telemedicine approaches to DF disease treatment is still in its infancy, facing technical limitations and implementation issues. However, various approaches have demonstrated the effectiveness or feasibility for assessing, monitoring, preventing or treating DF disease, and they need confirmation through studies to have broader use in DF care, particularly for patients in remote areas.
